What Do Membranes Do? Thursday, Nov 24 2011
We know that membranes in cells are complicated, and we have a pretty good idea of what they look like (Figure 1). We also know that the plasma membrane, the membrane that is the edge of the cell, is the thing that makes the cell a cell. At the very least, it means the cell is a discrete entity, and not just a loose collection of cell parts in one place. This might seem a serious job on its own, and it is, but there is more.
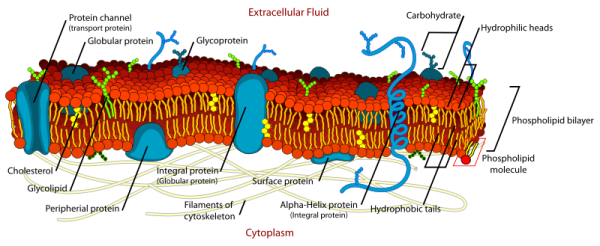
Figure 1. Schematic representation of a cell’s plasma membrane, showing the lipids along with peripheral and integral proteins, glycoproteins, surface proteins, ion channels, and carbohydrates. Note also the cytoskeleton filaments that, together with the plasma membrane of the cell, give it a life-supporting structure. Picture courtesy of WikimediaCommons.
Figure 2. Schematic representation of vesicles, with lipids forming a bilayer around a small body of water with other solutes (green blobs). Picture courtesy of WikimediaCommons.
Bilayers, such as that shown in Figure 1 contain various types of protein and some bits of carbohydrate*. However, to do the job of separating the cell’s insides from its environment, none of these other components are necessary. It is perfectly possible to create a sturdy bilayer from lipids alone. When this bilayer is formed in flat sheets and stacked up four-by-two, it is known as a lamellar phase [1], and when a bilayer is shaped like a ‘bubble’, it is known as a vesicle (Figure 2) [2]. Vesicles may sound a curiosity, but they are very useful in research laboratories. They provide a sort of stepping-stone to understanding complete cells as they can be of a similar size, but are much less complicated biochemically. So, if a scientist wants to test an idea or hypothesis without the complications of a testing it on a whole cell, a vesicular system is a useful one to try it on first. If the job of being a skin, a border between the inside of the cell and the outside, can be done so simply by a lipid bilayer, why does the cell go to the trouble of having the rest of the gubbins there?
The question becomes more pressing when we consider that there are a variety of proteins in the membranes of cells of both animals and plants. Not only that but, proteins are oten more complicated to make than lipids. The question has been raised not only about cells, but also about the parts of a cell that have a membrane or mono-layer covering of lipids. These parts of a cell are known as organelles.
Murphy and Cummins [3] asked this question directly about the organelles that store oil in seeds like Brassica Napa (rape seed). It is an interesting question, and there is an understood answer. Consider this: if you were to put together an organisation of people in order to produce, say, a new range of chocolates, you would undoubtedly want several types of person. You would want people who could physically make the chocolate itself, so that you had chocolate to sell; you would want people who could design the chocolates, so the product had a chance of being consistent, coherent and tasting good; you would want managers to over-see the work of both of these teams; you would want market analysts, a legal team, HR and so on. Many types of people, probably too tedious to name individually. But you would need something else, too. You would want them to be able to talk to one another. You, as the philanthropist of your very own chocolate–making enterprise, would need them to communicate so the company’s activity is co-ordinated and coherent. With cells, it is just the same: the various organelles need to be able to communicate with one another in order that the cell can stay alive. In order for a cell to stay alive, it must do a multitude of things – take in food, get rid of waste, resist changes in the environment, grow and replicate, to name but a few. And of course all of this must be co-ordinated so the cell does not die. An important part of this is cellular signalling.
As you might imagine, in order to perform a complicated set of processes, cellular signalling takes on a variety of forms – just like the large vocabulary required for a whole company to communicate with itself effectively. The complexity of cellular signalling has been made clear in recent decades, and now there are at least four scientific journals that focus on this aspect of biology alone, namely Cellular Signalling, the Journal of Cell Communication and Signalling, the Journal of Molecular Signalling, and Cell Signalling Biology. The role of lipids in cellular signalling is a wide topic, and will be examined in other entries in this blog. Examples include how insulin lowers blood sugar level, and how the cytoskeleton (Figure 1), and thus the shape of the cell, is managed.
References and Notes
*Anything with the prefix ‘glyco-’ either is a carbohydrate or has one attached to it.
[1] F. D. Gunstone, John L. Harwood, Fred B. Padley, The Lipid Handbook, 2nd Ed, 1994, ISBN-13: 978-0412433207.
[2] J. C. Stachowiak, D. L. Richmond, T. H. Li, F. Brochard-Wyart, D. A. Fletcher, Lab on a Chip, 2009, 9, 2003–2009.
[3] D. J. Murphy, I. Cummins, Phytochemistry, 1989, 28, 2063-2069. http://www.sciencedirect.com/science/article/pii/S0031942200979214