“Bubbles, Bubbles, Everywhere, But Not a Drop to Drink”
Soap is something we all use every day, without even really thinking about it. Well, most of us use it every day. But how much do you think about it? Do you feel you know what it does or how it works? Do you even need to know either of these things?
Of course it is perfectly possible to live a full and active life without knowing anything about soap at all. Most people get as far as which one(s) they like the smell of, and possibly which ones their skin does and does not react well to. What might help with the latter, as well as the behind-the-label study of these products, would be some understanding of the terminology used on these toiletries. For example, Pears® now market something for washing hands that is “100% Soap-free”. Bayliss and Harding market a “cleansing hand wash”, that is not soap free.
In order to understand the difference between these two, and believe me they both wash your hands perfectly well, we need to know what soap is, and what it does. It may seem trivial, but I would also suggest we need to know what its purpose is as well, so the stuff we rub all over ourselves in the bath does what we want it to.
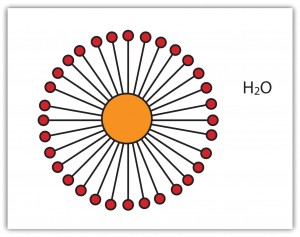 Figure 1. A cross-section of a micelle of the type formed by surfactants. The orange region (middle) is the grease around which the surfactant molecules are packed. Photo copyright: www.flatworldknowledge.com.
Figure 1. A cross-section of a micelle of the type formed by surfactants. The orange region (middle) is the grease around which the surfactant molecules are packed. Photo copyright: www.flatworldknowledge.com.
The obvious job for soap is to clean us, our clothes, our house/office etc., one way or another. What it is doing of course is removing grease and dirt. It does this by making the grease and dirt soluble in water. If you are thinking either that something like a lipid could perform this task, or, that such a mixture of grease and water is an emulsion, you are absolutely right. Tragically, although lipids are good at dissolving grease, they are often hard to wash away completely with just water. So we need something slightly different for this job. We need something that is more like a surfactant. As it happens surfactants and lipids are quite similar. They both have sections that like water (hydrophilic), and sections that like grease (lipophilic). However, the self-assembly properties of surfactants are slightly different to those of lipids. Typically, lipids form bilayers or cylinders, where surfactants form micelles (Figure 1). Micelles are particularly good at trapping grease as the surfactant molecules can fit around the grease droplets, with their water-liking head groups facing out. This allows the grease to dissolve in the water and therefore be washed away.
So we know what these sorts of molecules are doing physically. But what is soap? Well, like lipids and surfactants, in order to dissolve grease into water and form an emulsion, it needs aspects of both water and grease to be an effective emulsifier. Soap is in fact made from fat, known to scientists as fatty acids. The fat is boiled up with some caustic soda (sodium hydroxide), and so we have made a sodium salt from the fatty acid (Figure 2). This is more or less how soap has been made at least as far back as Tudor England. The source of fat in that case was mutton, and the hydroxide was cobbled together from heating mens’ urine. These days, sodium hydroxide is commercially available, and a variety of sources of the fat are used – everything from whale blubber to olive oil.
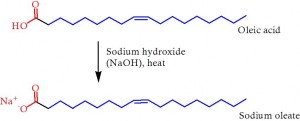 Figure 2. The preparation of soap from a fatty acid, using sodium hydroxide. This is essentially the same chemical process that has been used to make soap since Tudor times, and is also how it is apparently made in the film Fight Club. In the film, the main character uses fat from liposuction patients to make cakes of soap that he then sells to reputable shops.
Figure 2. The preparation of soap from a fatty acid, using sodium hydroxide. This is essentially the same chemical process that has been used to make soap since Tudor times, and is also how it is apparently made in the film Fight Club. In the film, the main character uses fat from liposuction patients to make cakes of soap that he then sells to reputable shops.
So that is not only the physics but chemistry of soap covered. One last thing you may like to know: how can we have soap-less soap? Well, ‘hand wash’. Lateral thinking would suggest that we need a surfactant that is not made from a fatty acid. And that is more-or-less what we use. The most common one is usually called sodium lauryl sulphate on packaging, though there are a couple of different names that are similar to that one. It is commonly used in shampoo. Scientists call it sodium dodecyl sulphate and it has many uses. For example, is used by molecular biologists to separate proteins according to their size. Think of that next time you are washing your hair.