Photocredit: Helen Zaltzman (with permission, @helenzaltzman)
The front page of Ms. Magazine this quarter sends a clear message. I saw it thanks to a tweet by Helen Zaltzman (picture). It is a message that the feminist movement, and particularly its hard-line wing, have been issuing for a long time. It indicates a long-standing problem.
It was interesting for another reason, too. The tweet was prescient as it came in the same week as Carrie Gracie’s resignation as the BBC’s China editor. A number of sources have quoted her as saying words to the effect that she did not want to collude in a system that paid women less than their male counterparts for the same work. Ms Gracie has made an extraordinary and commendable move. It’s also a disappointment, though. Why does someone senior have to resign to blow the whistle about activity that is, at least in legal theory, outlawed? It’s 2017. The equal pay act received Royal assent in 1970.
What’s also disappointing is how the self-sacrifice and the illegal nature of this are lost. Yes, the issue has been raised and yes it remains annoying even for male feminists like me who want to see a meritocracy. But it doesn’t stop there. What about the problems that don’t have the opportunity for leverage like a high profile resignation and an open letter? How can those problems be understood, and solved?
One of those problems comes from comparisons made in the pay gaps in different companies. One that has been quoted is a pay gap between women and men at a budget airline. I’ll call it SqueezyJet. Sources differ on the percentage, but all quotes are well into double figures for the difference between men’s pay and women’s at SqueezyJet. A bit of digging shows that the men and the women in the company are being compared to each other. This seems reasonable you may say, but then we find out that the cockpit crew are (surprisingly) overwhelmingly male and the cabin crew are majority female. Is it really a surprise that pilots and the people charged with passenger loading and unloading and schilling sandwiches are paid the same? I don’t think so. For me it is self-evident that women can be just as good at flying an aircraft as men. The fact that piloting is male-dominated hints at a problem with skills or training or recruitment, not pay. The real problem there has not only been missed but also conflated with something rather different.
Another exaple involves a job where a pay difference is as good as impossible. It is that of minimum wage jobs. In the late 90s I was doing my A-levels. I had a part time job, as did most of my friends. We wanted to feel grown up and independent but also we wanted a few quid for alcohol, sweets, books, condoms and even petrol. Two of my female friends worked at a local supermarket. I’ll call it Osda to hide its identity. Osda paid all its junior staff the minimum wage. The minimum wage had just arrived in Britain and so this was seen as basically fine. It seemed fair. However, it quickly became apparent that the work they were doing was not quite so fair.
The company would gather in enough junior staff or as many as they could and then apportion the jobs as they needed to on the day. My 17-18 year old female contemporaries were put on the tills. They didn’t really mind this to begin with—easy enough job, get to sit down, not cold in the winter, etc. However, they never got to drive the fork lift when the new stock arrived. That job always went to the same one or two people. The same one or two hairy, tubby, smelly men. But why? My friends had passed their driving test. They were qualified. They drove to work. Why weren’t they driving the forklift once in a while?
These examples—and I have no doubt there are countless other specific examples that you’d never see reflected in pay statistics—show that the ‘wage gap’ is being used to refer to a multitude of problems, not just one in which the salary two people are paid for the same job. It’s becoming a rather glib term.
That’s not to say the problems are glib or unworthy of our attention. They are not short-lived either. A slightly reluctant Mariella Frostrup said on the Today programme that the same things were happening in the 1970s. So these hidden problems are not even a new thing. This suggests to me that the current tactics and approach we are using to tackle these iniquities—anger and protest—are not working. The solution is partly a need for clarity about what the problems are, a need to inspire confidence and ambition, but also to focus on the intellectual case, and not anger.
]]>Diagnostics has been a central part of clinical medicine right from the beginning. It’s not hard to see why—how can a patient be treated properly without a clear idea of what the condition is? Diagnosing the problem is so fundamental that even the cranks and the nutters must also pay some sort of deference to it. It follows that the more precisely the condition can be diagnosed, the better crafted and thus more personalised, the treatment can be. Our understanding of the workings of human body has been established at a molecular level, as has our understanding of some disease.
The simplest forms of these are well known: the presence of a given metabolite in the blood or urine are obvious examples. Conditions such as diabetes and even pregnancy can be identified readily. These two are relatively straight forward to diagnose using metabolites: the presence of really any concentration of glucose or human chorionic gonadotrophin (hCG) is sufficient to indicate a positive identification.
However, not all conditions are similarly black-or-white. Some systems depend upon a development of tissues in order to function properly, for example in lungs. In order for a baby to be born and breathe properly thereafter, its lungs must of course develop sufficiently. However, assessing this is a challenge; how mature does a lung need to be in order to function? This is an important question as of course there is little time to do anything about poor lung function once the baby has been born.
A recent collaboration between several laboratories in Scandinavia and the UK has produced a way of testing for lung maturity before it is too late, by profiling the lipid composition of gastric aspirates [1]. This has allowed prediction of Respiratory Distress Syndrome with a high degree of accuracy, that may yet be used clinically.
A quick medical response is also required with many head injuries. Such injuries are well-known in contact sports such as ice hockey, but conditions such as concussion can be difficult to diagnose in the early stages, despite long-term neurological consequences for the patient. A recent study in Canada showed that the abundance of isoforms of PC are modulated in at least 90% of cases of concussion [2]. This offers an opportunity for both an additional diagnostic test and also a means for understanding how cell structure may change as a result of such an injury.
A third recent development may also assist in diagnosing injury, though it is one that can vary enormously between individuals. Lipid profiling has been used to predict clinical outcomes in patients suffering from burns [3]. It appears that increases in the abundance of unsaturated fatty acids in blood plasma correlate with death of the patient. It is not yet clear what preventative measures can be taken with these data, however, it seems that like the lipid profiling of gastric aspiration, lipid profiling of burns victims may give an insight that a visual examination may not and thus inform medics about the need for preventative treatment.
That lipid profiling may be used as a diagnostic tool in three quite separate conditions hints that it may be possible to use it in a host of other conditions, if only we know which lipids or lipid ratios to look at. These three studies have all required a healthy dose of serendipity and scientific intuition—as well as hard work—to find out what they have. We may not need to rely upon this nous to find all such examples, however. Identification of shifts in the lipid fraction that provide diagnostic evidence for other conditions may be informed by the availability of novel techniques that allow us to understand which lipids are where [4] and deeper analyses of lipid distribution [5].
References
[1] H. Verder, C. Heiring, H. Clark, D. Sweet, T. E. Jessen, F. Ebbesen, L. J. Björklund, B. Andreasson, L. Bender, A. Bertelsen, M. Dahl, C. Eschen, J. Fenger-Grøn, S. F. Hoffmann, A. Höskuldsson, M. Bruusgaard-Mouritsen, F. Lundberg, A. D. Postle, P. Schousboe, P. Schmidt, H. Stanchev, L. Sørensen. Acta Pædiatrica, 2016, DOI: 10.1111/apa.13683.
[2] M. Daley, G. Dekaban, R. Bartha, A. Brown, T. Charyk Stewart, T. Doherty, L. Fischer, J. Holmes, R. S. Menon, C. A. Rupar, J. K. Shoemaker, D. D. Fraser. Metabolomics, 2016, DOI: 10.1007/s11306-016-1131-5.
[3] P. Qi, A. Abdullahi, M. Stanojcic, D. Patsouris, M. G. Jeschke. Scientific Reports, 2016, DOI: 10.1038/srep38707.
[4] G. Lia, J. H. Kima, Z. Huanga, J. R. St. Claira, D. A. Browna, E. London. Proceedings of the National Academy of Sciences, 2016, DOI: 10.1073/pnas.1610705113.
[5] C. L. Jackson, L. Walch, J. M. Verbavatz. Developmental Cell, 2016, DOI: 10.1016/j.devcel.2016.09.030.
]]>
A recurring philosophical question is what makes us human. It’s a good question because it appeals to our understanding of our sense of self. Part of the answer is straightforward, like how we differ from inanimate objects and even other life forms like insects or trees. What separates us from other primates on the other hand, is a more difficult to pin down.
There are a number of well supported facts—humans are generally a lot less hairy than other primates, spend far more calories in completing any given task, walk on two legs and have the power of speech. Humans also have the lowest sperm count but the largest penis size of the primates.
However, these facts whilst interesting, are more a random assortment or cluster than a coherent argument or case for what human is. Can I hope to do better? Well, I have a suggestion for you, at least from a molecular point of view.
An early view of human development was that we evolved on the savannahs of Africa. The fossil evidence is certainly consistent with Africa being the cradle of humanity, however more recently, the savannah hypothesis has been replaced by one involving water. Evidence collected since the theory was proposed by Sir Alister Hardy in 1960 suggests that humans evolved from primates who lived near water*. Not only did the water provide a convenient supply of food, but also led to our upright movement, relative loss of hair, the development of subcutaneous fat, but also our lipid intake was different.
The evidence suggests that we ate fish, mainly giant catfish, in large quantities. The fatty acid profile of triglycerides in fish, especially cold water fish, is different to that of mammals in that they have more unsaturated bonds. In particular the fatty acids known as omega-3s, a category named after the position of the double bonds (See Fig. 1), such as docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA) were in rich supply. This was useful for brain development—an important part of how human cognition developed. We needed to be clever to follow the behaviour of animals to exploit them for our nutritional intake, to manage our intake throughout the year, but also the dexterity to catch such animals and make the best of the plant material available. This led to the development of a larger brain.
The idea that such a large brain could have developed on the savannahs seems possible and has also been supported—if humans eat the brains of other animals, for example. However, the supply of the omega-3 fatty acids is much weaker there, where that of protein is much higher. This leans away from humans and more towards musclier animals with smaller brains—indeed, animals with a similar brain/body size ratio as the prey they hunt.
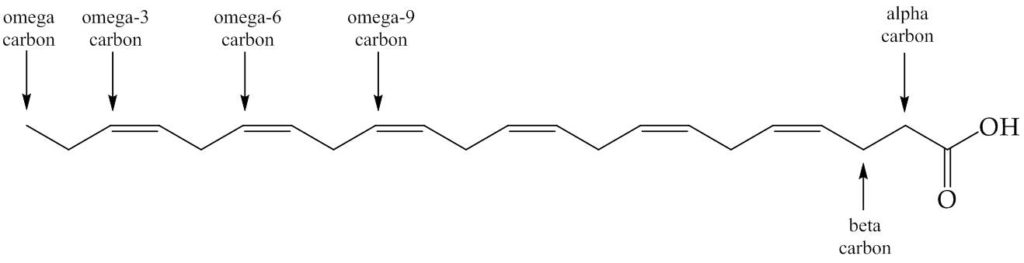
Figure 1. Docosahexaenoic acid, DHA. This fatty acid has twenty carbons and six cis-double bonds. The names given to the carbons are marked. The fatty acids referred to as ‘omega-3s’ are ones with a double bond at that position (includes DHA). The fatty acids referred to as omega-6s do not have a double bond at this position (includes linoleic acid).
The supply of omega-3 fatty acids is considerably higher in mammals in marine systems, enabling brains to enlarge. The comparisons are striking: the brain of a savannah mammal like a zebra is about 300 g, but that of a marine mammal of comparable size such as a dolphin, is about 1800g. This suggests that the supply of brain-building materials is much richer in food from marine environments. This analysis therefore supports the hypothesis that early humans developed larger brains partly because of the access to marine livestock. It is also consistent with the evidence that our brains perform better when we eat animal and especially marine animal, rather than plant, unsaturated fat.
The accumulation of omega-3 polyunsaturated fats by a land animal is therefore much more ‘human’ than one might have thought. The uniquely-human aspect may not end there—part of the grisly evidence that led to the conviction and hanging of John George Haigh, the acid bath killer, was a considerable quantity of human fat that was not destroyed by the sulfuric acid he used. Even with primitive techniques it was possible to identify it.
This analysis has one other intriguing possibility. If correct, it suggests that humans moved from in-land areas towards water, and whilst there was some contact with it, we remained land-based animals. Did any such primates develop further, or even leave the land altogether?
We may never know for certain, and we have more answers and more questions as a result of this work. We also have a better idea of where we might fit in the world and can therefore infer what it is to be human from the evidence of the environment that may have produced us.
References and further reading
*Much of the background to these ideas is explained beautifully in a radio programme by Sir David Attenborough that can be found at http://www.bbc.co.uk/programmes/b07v2ysg . This programme is also a great joy for a lipid scientist pedant as the distinction between lipid and fat is made clear, as is even the type of fatty acid.
]]>
Biology is an inspiring subject. The variety of living organisms, from the smallest bacterium to the largest Sequoia tree, to processes such as birth that are often described as miraculous, have captivated intellectual and poetic interest since ancient times.
And it isn’t just the outside of living organisms and the systems of which they form part that amaze us. The human heart can pump blood continuously for over a century. Ants that weigh no more than a few hundred milligrammes can build coherent structures that weigh hundreds of kilogrammes each. We currently share the Earth with the largest animal ever to have existed, the blue whale, that gives birth to young that are typically over two tonnes in weight after a gestation of a little over eleven months.
The fundamental processes on which these spectacles rely have been gone into in some detail. We know much about how the heart works, and how ants move sand and soil around. Studies of fertility and the cell cycle have told us about where babies come from. This satisfies much of our curiosity, but some questions remain. For example, how can a cell that will go on to become a blue whale or a person, prepare itself for being broken in half billions of times in succession to create an individual?
This question is not as sarcastic or as niche as it may sound. In order to get from one cell to two, and thus from a fertilised ovum into a healthy baby, cells must divide. This is described as binary fission (bacteria) or cytokinesis (mammalian cells), but it amounts to the same thing: the controlled partitioning of one cell to leave two, living cells. There are bacteria that can do this every twenty minutes, and the billions of cells in a newly-born blue whale have all grown from just one cell, a fertilised ovum.
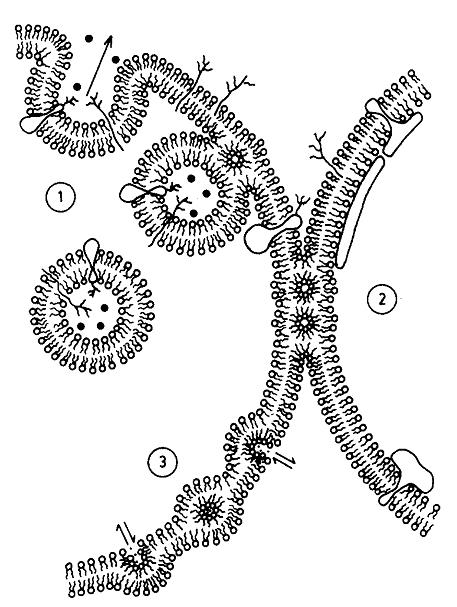
Figure, reproduced from Cullis et al. [6]. Original text: A metamorphic mosaic model of biological membranes illustrating various aspects of membrane morphology and function potentially involving non-bilayer lipid structure. In region (1) an exocytotic fusion event proceeding via an intermediate inverted micellar or inverted cylinder organization is shown, whereas in region (2), inverted cylinder structure allows a stable semi-fused inter-bilayer connection to exist, possibly corresponding to tight junctions. In region (3) enhanced permeability to divalent cations is proposed to proceed via an inverted micellar intermediate, which may correspond to the ability of phosphatidic acid to act as a Ca++ ionophore.
Recent work by Keidel et al. [1] and Zhao et al. [2] investigated both membrane scission and fusion. Insodoing, they have provided evidence for how the membrane part of cell division proceeds. The data published provide evidence for hemi-fission and hemi-fusion intermediates, one in which the inner monolayer of the membrane is broken first, followed by the outer monolayer. This is consistent with an hypothesis generated in biophysics some time ago (Figure), and so this has probably lifted a scientific purgatory.
However, further challenges in understanding the structural changes in the cell cycle remain. The limited window for the light-dependent part of photosynthesis means that several species of algae time their cell division around light and dark periods [3, 4]. Furthermore, under favourable conditions, several species of algae are able to undergo so called multi-fusion [5]. This means in effect breaking one cell into as many as sixteen daughter cells. It’s a huge structural task that must happen in as controlled and consistent a manner as binary fission, in order not to kill the cells that attempt it. Multi-fission must also work around the division of chloroplasts so that all of the daughter cells have enough chloroplasts to make best use of the light by the next light period commences.
Further work is required to understand these, however study of the algal cell cycle is tantalising. It may yet shed light on binary fission, its speed and limits, and indeed how the whole process of cell division has evolved.
References
- A. Keidel, T. F. Bartsch and E.-L. Florin, Scientific Reports, 2016, 6, 23691. 10.1038/srep23691. http://www.nature.com/articles/srep23691#supplementary-information.
- W.-D. Zhao, E. Hamid, W. Shin, P. J. Wen, E. S. Krystofiak, S. A. Villarreal, H.-C. Chiang, B. Kachar and L.-G. Wu, Nature, 2016, advance online publication. 10.1038/nature18598. http://www.nature.com/nature/journal/vaop/ncurrent/abs/nature18598.html#supplementary-information.
- M. Vítová, K. Bišová, D. Umysová, M. Hlavová, S. Kawano, V. Zachleder and M. Čížková, Planta, 2010, 233, 75-86. 10.1007/s00425-010-1282-y.
- V. Zachleder, K. Bišová, M. Vítová, S. Kubín and J. Hendrychová, European Journal of Phycology, 2002, 37, 361-371. doi:10.1017/S0967026202003815.
- V. Zachleder, K. Bišová and M. Vítová, in The Physiology of Microalgae, eds. A. M. Borowitzka, J. Beardall and A. J. Raven, Springer International Publishing, Cham, 2016, pp. 3-46.
- P. R. Cullis, M. J. Hope and C. P. S. Tilcock, Chemistry and Physics of Lipids, 1986, 40, 127-144. http://dx.doi.org/10.1016/0009-3084(86)90067-8.
]]>
When Baker et al. quietly published a gender-based difference in the concentration of lyso-phosphatidic acid (lyso-PA) in 2001 [1], it wasn’t taken all that seriously. This was partly because the methods they used for both isolating the lipid fraction and for profiling it were not conventional, and partly because it was quite a narrow result in the grander scheme of things. However, recent work by Sales et al. [2] might just be enough to broaden this observation. Sales et al. found that not only is there probably about a 10% natural variation in the lipid profile in healthy individuals, but that the profile of both lipids and fats is related to gender, the use of hormonal contraceptives and personal disposition.
Sales et al. report that there is a statistically significant difference in the blood plasma concentration of several lipids. This includes similar lyso-lipids to the lyso-PA targeted in the Baker study, but also larger fraction, mainstream lipids such as PC, PE, PI and sphingomyelin (SM) in healthy men and women in their 20s.
The differences between healthy women taking hormonal contraceptives and those who do not also appear to be significant, and they are also significantly different to the men. At first sight, the variation within each group is wider than the variation between groups, but student t-tests indicate that the likelihood of the difference in absolute values being down to chance is less than 5%, indicating a significant difference.
There is also evidence that the concentration of certain glycerides, including fats, is partly gender- and hormonal-contraceptive-dependent, with members of the latter group having higher triglyceride concentration than women who do not take hormonal contraceptives. This may have implications for treating illnesses related to lipoproteins, though further work is required to determine whether gender or hormonal contraceptives have a significant role in this.
This work is interesting enough on its own, but it comes shortly after another study, by Aviram et al., that showed that lipid distribution in certain parts of cells varies during the day [3] and a third, by Dawaliby et al. that showed that a lipid called PE is an important regulator of membrane fluidity in eukaryotic cells [4]. These studies between them show that the concentration of major structural lipids such as PE and PC vary throughout the day and are dependent on your gender and the pills you take.
These studies are juicy for lipid researchers because they support notions that lipid geeks like me have worked with for some time. For example, that the lipids we require for our cells to function are a dynamic and responsive group of molecules that we have yet to fully understand.
This work also raises a variety of questions. Are other factors important, like diet, temperature, the seasons etc.? What about the difference between the healthy systems studied and metabolic diseases? The list goes on. It also hints that lipids are under-rated parts of our cells that might be able to give away more information than we think.
References
1. D. L. Baker, D. M. Desiderio, D. D. Miller, B. Tolley and G. J. Tigyi, Analytical Biochemistry, 2001, 292, 287-295. 10.1006/abio.2001.5063.
2. S. Sales, J. Graessler, S. Ciucci, R. Al-Atrib, T. Vihervaara, K. Schuhmann, D. Kauhanen, M. Sysi-Aho, S. R. Bornstein, M. Bickle, C. V. Cannistraci, K. Ekroos and A. Shevchenko, Scientific Reports, 2016, 6, 27710. 10.1038/srep27710.
3. R. Aviram, G. Manella, N. Kopelman, A. Neufeld-Cohen, Z. Zwighaft, M. Elimelech, Y. Adamovich, M. Golik, C. Wang, X. Han and G. Asher, Molecular Cell, 2016, 62, 636-648. 10.1016/j.molcel.2016.04.002.
4. R. Dawaliby, C. Trubbia, C. Delporte, C. Noyon, J.-M. Ruysschaert, P. Van Antwerpen and C. Govaerts, Journal of Biological Chemistry, 2016, 291, 3658-3667. 10.1074/jbc.M115.706523.
Pretty much everyone has heard of omega-3s. Health geeks will probably be able to go further and name DHA and EPA as omega-3 fatty acids. It is also well-known that these are poly-unsaturated fatty acids. But why should these be more special than others? And are those others therefore bad? And why are there different ones anyway?
The answer to last of those questions is that fatty acids come from several different sources—plants, fish, mammals, birds—that live in different environments and thus that have different requirements of their fats. This is reflected in the molecular structure of the fatty acids, and thus which sort they are.
The answer to the second question is that is that no fatty acid is intrinsically bad as such, though if we consume more of them than we use the result is an increase in the volume of fat stores, leading to obesity in the long term. So, there is a limit to what we need regarding fat.
The answer to the first question is that there are some fatty acids we need for certain things that others cannot really substitute for. DHA is well-characterised in this regard. For example, there is evidence that it has a role in human behaviour1, 2. Adolescent children who are at risk from bipolar disorder have lower levels of DHA and EPA1, as do younger children with ADHD2.
It does not stop with children who are already unwell. There is mounting evidence that these poly-unsaturated fatty acids have a role in cognition not only in children3, 4, but also in adults with dementia5. Evidence drawn from groups of a wider age span suggest that it affects cognition throughout life6.
So, one might conclude that a supplement of DHA and EPA and we can knock ADHD, bipolar disorder, dementia and being a bit crap at spellings tests or the crossword, on the head. This would be easy to organise; such supplements are amongst the most easily available of any that are commercially available. Sadly, this is naïve. However, it is not immediately obvious why this should be.
Evidence has begun to emerge that not all children who have plentiful DHA and EPA in their diets exhibit the same good cognition as others who have the same amount but live elsewhere. This encouraged investigators to dig deeper. One factor that has appeared is that people who live in places where a higher fat diet is more common, tend not to exhibit the cognitive benefit of DHA and EPA. One study in particular was conducted with populations from 28 countries, taking into account other factors such as wealth as well, showed this correlation neatly3. This work concluded that omega-6 fatty acids in effect drowned out the omega-3s, suggesting that the right ratio of the two would give good results.
But what is the magic ratio? This has also been researched3, 7, 8. It seems that humans evolved for a diet of a ratio of about 1:1 of omega-6 to omega-39, though up to about twice as much omega-6 as omega-3 is still regarded as optimum. This can also be written as a ratio of up to 2:1 linoleic acid to the sum of DHA and EPA. Some of the western diets investigated showed a ratio of around 50:110 with an average of around 16:19.
This invites a second level of finger wagging from dieting know-it-alls: not only should we eat less fat in order to tackle obesity*, but virtually no omega-6 fatty acids either. In theory, this should be easy to implement. Our bodies are able to make saturated fat and can produce unsaturated fats from saturated ones with enzymes called desaturases. We only really need things like DHA and EPA in our diet because in practice we cannot really make them.
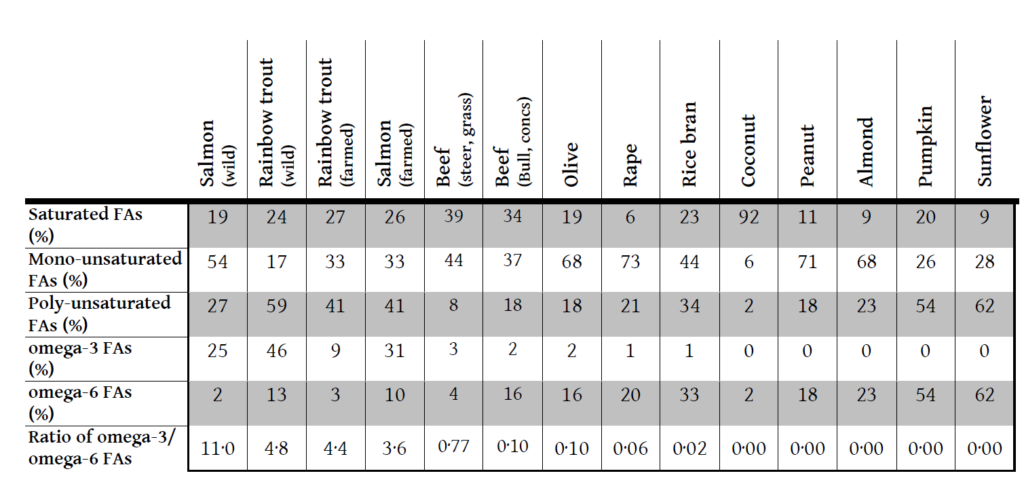
This theory is helpful, but once again the next step is not necessarily intuitive. Which foods fit into this mould and which do not? The table below shows data on the fatty acid profile of oils from various foods11-13.
I think I will have salmon for lunch. And hold on the sunflower oil in the mayonnaise.
References
*Some dieticians, quacks and faddists argue that (bang on about) a low carbohydrate diet is a good way of losing weight. Lowering carbohydrate intake can reduce the calorific intake. However, it contains less than half the number of calories per gramme as fat. So if an individual ingests more calories than they use, lower carbohydrate intake may be a less strong way to reduce overall calorific intake.
1. R. K. McNamara, R. Jandacek, P. Tso, T. J. Blom, J. A. Welge, J. R. Strawn, C. M. Adler, S. M. Strakowski and M. P. DelBello, Early Intervention in Psychiatry, 2016, 10, 203-211.
2. A. Crippa, C. Agostoni, M. Mauri, M. Molteni and M. Nobile, Journal of Attention Disorders, 2016.
3. W. D. Lassek and S. J. C. Gaulin, Prostaglandins, Leukotrienes and Essential Fatty Acids (PLEFA), 2014, 91, 195-201.
4.K. W. Sheppard and and C. L. Cheatham, The American Journal of Clinical Nutrition, 2013, 98, 659-667.
5. M. Loef and H. Walach, Journal of Nutrition in Gerontology and Geriatrics, 2013, 32, 1-23.
6. M. Weiser, C. Butt and M. Mohajeri, Nutrients, 2016, 8, 99.
7. C. Hoyos, C. Almqvist, F. Garden, W. Xuan, W. H. Oddy, G. B. Marks and K. L. Webb, Asia Pac J Clin Nutr, 2008, 17, 552-557.
8. S. Yehuda and R. L. Carasso, Proceedings of the National Academy of Sciences of the United States of America, 1993, 90, 10345-10349.
9. A. P. Simopoulos, Biomedicine & Pharmacotherapy, 2002, 56, 365-379.
10. M. A. Martin, W. D. Lassek, S. J. C. Gaulin, R. W. Evans, J. G. Woo, S. R. Geraghty, B. S. Davidson, A. L. Morrow, H. S. Kaplan and M. D. Gurven, Maternal & Child Nutrition, 2012, 8, 404-418.
11. C. Blanchet, M. Lucas, P. Julien, R. Morin, S. Gingras and É. Dewailly, Lipids, 40, 529-531.
12. M. Enser, K. G. Hallett, B. Hewett, G. A. J. Fursey, J. D. Wood and G. Harrington, Meat Science, 1998, 49, 329-341.
13. J. Orsavova, L. Misurcova, J. Vavra Ambrozova, R. Vicha and J. Mlcek, International Journal of Molecular Sciences, 2015, 16, 12871-12890.
]]>
I first heard about Alzheimer’s Disease (AD) when I was about nine. I remember the troubling feeling of how the condition takes hold, of how sufferers become a warped shadow of their previous selves. Since then, AD has become more common. This is mainly because the occurrence of heart disease and cancers, that kill humans earlier, have begun to fall. This apparent increase in AD has motivated funding bodies to grant money to research focused on AD and related conditions.
Early examination of the corpses of patients with dementia found that most of the body was quite normal. The damage appeared to be limited to the Central Nervous System (Brain and spine), where there were abnormal and typically rather long fibres. Initially, these were thought to be made of starch, but it quickly became apparent that they were proteinaceous and the more recently, a mis-folded protein. It’s not hard to guess that a build-up of a large amount of wrongly-built protein might get in the way of normal cellular activity. Perhaps unsurprisingly, it also leads to cell death. This loss of cells in the brain fits well with the decrease in cognitive function. This decrease in ability to process information and remember things can be acute; the test does not look challenging to someone with normal cognition [1].
Since AD has become more common, its subtleties have begun to emerge. For example, there are now well-recognised early- and late-onset types. The early onset is regarded as familial because it is associated with inherited faulty genes. Late-onset AD is associated with the loss of function of a different protein, called ABCA7. This protein is a lipid transporter, and is therefore part of the system that moves lipids around the cell.
Moving lipids about the cell is useful because it ensures that the right ones are in the right place, but also means that the right lipids are made. For example, cells that cannot transport PS to the mitochondria are entirely unable to make PE. Not being able to make or transport PE can be a real problem for the cell. For example, work completed in Japan over about three decades, showed that PE probably has a crucial structural role in cell division [2-7]. A cell that was unable to move it would not be able to divide.
Some very recent work has also shown that PE is an important component for ensuring the membrane has the correct physical properties for its function [8]. Furthermore, a study by Sakae et al. [9], that researched the lipid profile of mice who do not have working ABCA7 transporters showed that the amount of PE was about 36% lower in affected mice, against the control group. As PE typically represents about 30% of the membrane, this may also represent an effective increase in the abundance of other lipids. This may therefore effect a considerable change in the physical behaviour of the membrane, as the concentration of virtually all of the components will be changed.
This change in the lipid composition correlated with the type of memory loss observed in AD [9] and represents a nice insight into what role lipids may have in brain and spinal cord nerve activity. The broader question now is, if this effect can alter the function of membranes so much, what would a smaller change do? Effects of perhaps 75% activity of this transporter may be observable over a lifetime. Certain diets or malnutrition may mean that particular membrane components may be less abundant or absent, leading to a significant difference in the physical behaviour of the membrane and therefore the cell.
References
[1] S. Srinivasan, Neurology India, 2010, 58, 702. DOI: 10.4103/0028-3886.72167.
[2] S. Y. Choung, T. Kobayashi, K. Takemoto, H. Ishitsuka and K. Inoue, Biochimica et Biophysica Acta (BBA) – Biomembranes, 1988, 940, 180-187.
[3] K. Emoto, H. Inadome, Y. Kanaho, S. Narumiya and M. Umeda, Journal of Biological Chemistry, 2005, 280, 37901-37907.
[4] K. Emoto, T. Kobayashi, A. Yamaji, H. Aizawa, I. Yahara, K. Inoue and M. Umeda, Proceedings of the National Academy of Sciences, 1996, 93, 12867-12872.
[5] K. Emoto, O. Kuge, M. Nishijima and M. Umeda, Proceedings of the National Academy of Sciences, 1999, 96, 12400-12405.
[6] K. Emoto, N. Toyama-Sorimachi, H. Karasuyama, K. Inoue and M. Umeda, Experimental Cell Research, 1997, 232, 430-4.
[7] K. Emoto and M. Umeda, The Journal of Cell Biology, 2000, 149, 1215-1224.
[8] R. Dawaliby, C. Trubbia, C. Delporte, C. Noyon, J. M. Ruysschaert, P. Van Antwerpen, C. Govaerts, Journal of Biological Chemistry , 2016, 291, 3658–3667. DOI: 10.1074/jbc.M115.706523
[9] N. Sakae, C. C. Liu, M. Shinohara, J. Frisch-Daiello, L. Ma, Y. Yamazaki, M. Tachibana, L. Younkin, A. Kurti, M. M. Carrasquillo, F. Zou, D. Sevlever, G. Bisceglio, M.Gan, R. Fol, P. Knight, M. Wang, X. Han, J. D. Fryer, M. L. Fitzgerald, Y. Ohyagi, S. G. Younkin, G. Bu, T. Kanekiyo, The Journal of Neuroscience, 2016, 36, 3848 –3859.
]]>
A combination of more-or-less constant war, and the honour of winning it, created a desire for constant improvement to castles built in England in the middle ages. At first they were little more than a place to house soldiers. They rapidly gained status as the centre of local power—at first to protect the local people, and after the Norman invasion, to subdue them. Thus, attacking and taking castles became a way to gain power and destroy enemies. It therefore became useful to be able to break into a castle and destroy or replace the occupants, if not bring down the whole structure. Siege tactics were used sometimes, but impatience or an army equipped for violent attack led to more direct attacks on castles with a view to breaking into them. The latter had a stronger political effect as well as a more immediate shift of power.
There is a similar motivation that exists in the present day. However, these enemies are not the symbols or seats of power of bloodthirsty would-be usurper kings, but are equally fatal. And, like mediaeval fortifications, cancer cells cannot always be defeated by siege methods alone. Sometimes molecular invaders need to break the metaphorical walls of the castle, the cancerous cell’s membrane, in order to over throw the (biological) usurper who will otherwise bring the kingdom down.
There are two ways to do this. Either the biological soldiers (drug molecules) can break in through small and transient holes in the walls (cell membrane), or they can precipitate large-scale destruction of the walls (weaken the membrane so the cell falls apart). Both destroy the integrity of the castle, but in quite different ways.
In fact both of these approaches make good strategies for destroying cancer cells in human bodies. Cancer therapies can either interfere with the membrane such that it no longer provides protection for its cell, or they can be developed so that they pass through it easily and earn their honour by killing the cell from the inside. Exciting recent work from several groups explores both of these approaches, and provides useful basic information for possible future attacks. Neves et al. report evidence for the anti-cancer compound resveratrol interfering with the function of the cell membrane [1] –they change its structure and fluidity. Purushothaman et al. investigated the relationship between the rate at which anti-cancer drug norfloxacin passes through the membrane, and the lipids in that membrane [2]. Future tactics may be informed by work by Dawaliby et al., who have investigated the role of a particular lipid (phosphatidylethanolamine, PE) in directing the physical properties of the cell’s membrane [3]. However, the search is not yet over: much further work is required to understand the membrane perfectly with respect to drug interactions. So, like the Norman and Plantagenet kings in their search for victory and honour, our search for life after cancer, goes on.
References
[1] A. R. Neves, C. Nunes, H. Amenitsch, S. Reis, Soft Matter, 2016, 12, 2118. DOI: 10.1039/c5sm02905h
[2] S. Purushothaman, J. Cama, U. F. Keyser, Soft Matter, 2016, 12, 2135, DOI: 10.1039/c5sm02371h
[3] R. Dawaliby, C. Trubbia, C. Delporte, C. Noyon, J. M. Ruysschaert, P. Van Antwerpen, C. Govaerts, Journal of Biological Chemistry , 2016, 291, 3658–3667. DOI: 10.1074/jbc.M115.706523
]]>At least one senior tory has been accused of calling UKIP members ‘swivel-eyed loons’. It’s the sort of report I don’t want to follow up because it can only spoil the entertaining rudeness, but I definitely haven’t detected a massive backlash of denying that it was said, and furthermore, no one has said that it isn’t true. This suggests simultaneously that UKIPers are swivel-eyed loons and that everyone thinks they are.
If they are, by Jove there are a lot of them. They polled over 3m votes in the European elections in the UK in 2014, putting them in first place. They doubled their number of seats. Something like 4m people voted for them at the 2015 general election—about the same number as the Liberal Democrats and the SNP put together. They won one and came second in 120 seats. This odd result is probably inexperience at running a national campaign: they are a new party and so are not (yet) very co-ordinated.
But no amount of co-ordination can give a political party a win or even be noticed without solid underlying support. With the conservative party trying to unite and move more towards the centre ground, there has been less obvious Euro-scepticism from them. Support for the BNP has all but vanished. The support UKIP are enjoying is doubtless the result of a shift from both of these.
And they have a real chance of getting what they want. There is going to be a referendum on our relationship with Europe. UKIP, many conservative and ex-conservative MPs, and labour and ex-labour MPs, are calling for Britain’s Exit, known as Brexit. Britain’s age-old antipathy to Europe may yet be expressed by us leaving the EU in the next year or two. It probably requires co-ordination not just of UKIP but a lot of other, rather odd bedfellows, if they are to be sure of winning. But with that variety of political colours, there’s a good chance there is enough support for the No campaign and thus that we will leave the EU.
I feel slightly guilty about saying ‘we’ there. I am British and I remain proud of so being, however, I haven’t lived in Britain for nearly three years now and I don’t plan to move back for another two-and-a-half at least. I lived and worked the Netherlands from 2013-2015 and then in the autumn of 2015, started living and working in Norway. This gives me the lofty feeling that my opinion about EU stuff is informed; I have recent experience of one country at the heart of the EU and another that is not really involved but is also geographically near. In fairness, what it does amount to is one person’s experience of living in a proper EU country and (another) one that sort-of is.
In taking a point of view, I have tried to stack up what I see of the choices the governmental and corporate institutions have made, and use this as a way of measuring what being in the EU and nearly being in it means. In the Netherlands, the income tax is about 42% for everyone, and you have to buy private health insurance on top of that and are charged it backdated to the moment you arrive. The trains are cheaper than the UK, but not more numerous or more on time. All of the supermarkets have the same bland food, poor baking and limited range of fresh or cooked meat. One of the first things I saw of my local supermarket was a roll-up sign outside proudly announcing that it was going to begin opening on Sundays. They don’t yet accept internationally-recognised credit cards, though local debit cards are accepted (welcome to 1992). There are no parliamentary constituencies, so there is no being bothered by political campaigning but the government is typically a coalition. The current one is between their equivalent of Conservative and Labour parties, called VVD and PvdA respectively. They have a staggering number of bike lanes that I love to pieces, and meant I cycled something like 26,000 Km over the 29 months I lived there. Maybe the scandalous income tax pays for that, but somehow I doubt it.
Norway is an EEA country, by contrast, so ‘nearly’ EU. I have heard Norwegians described as social democratic politically, and this seems a fair representation. Another one is that they like consensus, which they do, not unlike the Dutch. Income tax for me here is 35%. Healthcare bills are also on top of income tax though there is no legal obligation to buy health insurance. The costs of healthcare are gnatty, just raising an invoice for a visit to the GP costs kr. 55 (about €5·50 or £4·50). And, as we have found, the trip may be wasted anyway as their reputation for under-medicating is well-founded. The importation of wine is state owned and controlled, and subject to monstrous import tariffs. At any rate, a bottle of Chablis costs about 50% more than it would do anywhere else in Europe and you can’t even buy the bloody thing in a supermarket. You can only buy it 1000-1700 Monday-Saturday from one or perhaps two places in the whole of the city in which I live. Beer and a few types of cider are available more readily, and from one of the limited range of dreary supermarkets and there are ones from other countries, but even a can (yup, a can) of cheap cider will cost about three quid. I’ve no idea where the tax is going; the roads are not good here, potholes you could buckle a wheel in or be thrown off entirely by, and the existence of bike lanes is often debateable at best. Roads do get salted and gritted, though, as everything would stop if they didn’t. Perhaps the fiscal income is low—Sunday opening exists only in the run up to Christmas here and you can’t buy alcohol after 2000 or at all on Sundays.
This rather depressing catalogue makes me think that things are actually managed rather well in Britain, or London at least, and that being in Europe or nearly out of it is no help to us. If income tax that is 50% or more higher than in Britain without much tangible benefit is what the consensus politics of closer ties to the EU means, then frankly it’s hard to come to the conclusion that it’s good for us. And it’s a pity they’ve failed on such big things, because the smaller successes—like how easy it is to start working in the EU as a British person (or having a British passport anyway), are a definite plus. But even the most avid pro-European can’t claim that the £87bn that the EU has cost over the last four years is really good value for money if the best it can do is lighten the admin load.
So, although I have enjoyed living in Europe, the fiscal structure in both the Netherlands and Norway means I can’t tell you that I have fallen in love with either the European dream or the nearly-European dream. But that’s just it, I am arguing for leaving the EU altogether on economic and statutory grounds. I wonder if someone who has never left Romford, Barnsley or Exeter, and who does vote for UKIP, uses the same arguments to justify their position.
The fact is that Brexit may happen, and if my experience is not way off, probably should. It would give us power over our own destiny, more cash that could be soent on something useful like scientific research, and no greater admin burden. But, if Brexit does come to pass, it will probably be thanks in no small part to the votes of swivel-eyed loons and their thinly-veiled racism. I’m in the peculiar position of agreeing with the expected vote of a lot of confirmed ‘no-to-the-EU’ types, a lot of whom are people I’d cross the street to avoid. Little surprise then, that the No campaign and not just the loons themselves, are uncoordinated. Let’s hope the UK won’t be after a ‘no’ win. At least at the moment we are united by all disliking the EU.
]]>I love being cited. The leap of excitement when I get an alert e-mail from google scholar telling me that someone has not only found and read my work, but decided to refer to it in theirs, has a long tail. I feel chipper all day. It doesn’t really matter to me whether it forms part of their argument for conducting their work, or informs their analysis, it’s all good as far as I am concerned. I feel rewarded and motivated to work harder.
It’s a nice upside to publishing. Getting the same sort of alert from a slower system and finding out it was a self-citation to paper published a year ago, doesn’t have the same effect. Neither does realising that a paper or review that’s been out for six months has not yet been cited. But these are both simple, if not slightly gloomy reflections. The ones that can really play with your mind, and make you write blog posts, are when it’s a bit more complicated.
A year or two ago, I got around to publishing some data that had been lying about for a while and was just too big a pile to ignore. Despite its size, I felt the paper came together well, and we gave it a clear title and got it into a respectable journal. I was tickled pink that it was cited within about three months of publication. But, curious type that I am, I wanted to see how they’d used it. I supposed that their research was potentially as relevant to me as mine was to them (or so I told myself). What I found was a surprised me. They’d misread my paper. My paper shouldn’t really have been cited at all. What they needed instead was a reference to a slightly different lipid.
I felt a bit guilty, if I am honest. It was a bit like the time I was given too much change in a busy shop on payday—I didn’t feel like I needed the cash, but somehow there was no opportunity to give it back. Unlike that situation, with the mis-referencing, I then felt a pang of annoyance. How could they get it wrong? After all effort I have made and bollockings I have had about correct referencing, I was reading an article in which they hadn’t bothered to read the title of the paper they were referring to.
What I have been telling myself since is that this is Taoist karma. Obviously in reality this is as much hogwash as the next thing, but hear me out. I regularly tweet about lipid research (I’ve run out of jokes that fit into <140 characters and had no desire to shut up altogether, clearly) and so I see the new literature that comes up in the general field concerning amphiphilic biomolecules. Importantly for this anecdote, this is also the area in which I publish.
Can you feel what’s coming? Yes, that’s right: the new literature I see includes the papers that could or should have cited my work, but didn’t. There was a particularly fine example this morning.
It gave me a similarly powerless feeling to the one of being given a citation which I felt I didn’t deserved. The only difference was that I had a greater sense of injustice about not getting a citation rather than getting one incorrectly. But, the net result in my citation count is the same. So should I mind?
]]>
Evolution can be defined as the modulation of a set of inherited characteristics of individuals by environmental conditions. In this definition, the characteristics that fit the compromise of the species and the environment best, are those that survive in a given species.
This applies to microbes, also called single-celled organisms, as well as much to larger, multi-cellular organisms such as humans. Perhaps partly because they are so small, and thus can be dispersed easily, microbes are more diverse than any other group of living organisms. They span two domains of life, archaea and the more familiar bacteria.
The theory of evolution explains the existence of this variety as the exposure of single-celled organisms to a great variety of conditions (virtually the whole of the Earth) over a considerable time scale (billions of years). We are therefore not at all sure we know what all of these microbes are. We are only tentatively sure of the extent of the environment in which they live. Archaea, the third domain of life sometimes called extremophiles, were formally described as late as the 1970s. New bacteria are discovered every day—some in the Amazon rainforest, and some in hospitals in which antibiotics have been prescribed too much.
Such a range of bacteria would not survive if they could not also live within certain changes in their own environment. Such changes include the seasons and thus wet and dry conditions, as well as warm and cold. These are important for the cells to cope with as they affect the availability of nutrients, oxygen and sunlight. They also affect the amount of warmth available. We associate this with comfort—it’s nicer to be in a warm place than a cold one, only a very few people seek to go somewhere cold on holiday purely for that reason. However, there is also a thermodynamic reason. Warmer rather than less warm is a change in the thermal energy available, something that changes the behaviour of the molecules that constitute cells.
When energy is lower, cells are typically less active. However, cells that survive are ones that can still live, and better still grow, under such conditions. Hardy microbes therefore have mechanisms for coping with the cold, that can be called upon when the temperature drops. Yeast is one such microbe. It has also found use as a research tool because it is a eukaryotic cell that has only about 5k genes and can be manipulated genetically and grown in a laboratory.
The form of yeast known as Saccharomyces cerevisiae, is the principal sort studied in laboratories. It has evolved the ability to grow under cold conditions. In fact, it has recently been discovered that a key part of this adaptation is the result of a single gene. Study of a strain of S. cerevisiae with a mutation of its inp51 gene have shown that a major signalling lipid called PIP2 is linked to both membrane fluidity and cell growth. This suggests that membrane fluidity is key to the survival of S. cerevisiae under cold conditions.
Córcoles-Sáez et al. showed that S. cerevisiae with the mutated inp51 gene had a lipid fraction around 40% smaller than that of the unmutated form (wild type), and moreover that the membrane that was there was less fluid [1]. This showed that this gene influences not only the size of the lipid fraction but also what it contains. The size of the fats fraction (triglycerides) was reduced still further, by around 68%. This implies that this gene is involved in the synthesis of all fatty-acid-containing biomolecules. This is because it affects the abundance of both energy storage molecules (fats) and structural molecules (lipids) that comprise fatty acids.
Although spectroscopic determination of the lipid profile has only been applied to the wild type S. cerevisiae [2], it seems clear from chromatographic data alone that there are differences between the two strains, especially in the larger bulk-lipid fractions such as PC and PE [2]. This underscores the fundamental nature of the shift to lipid metabolism.
This raises the question of what the mechanism is between this gene mutation and the lipid fraction. The inp51 gene encodes for a phosphatase (enzyme) that turns PIP2 into another lipid, PI-4-P. This enzyme therefore catalyses the removal of a phosphate, which has the biological effect of turning the PIP2 lipid signal off. This also means that PI-4-P is not made, the effects of this lipid are not observed, thus probably limiting the extent of stored curvature elastic stress [3]. This suggests that PIP2 restricts phospholipid synthesis, and thus may be of use during periods where the cell needs to change or perform some other function, rather than to grow or repair itself.
This represents an extraordinary level of influence for a single gene. The ability to turn off this lipid signal influences an entire fraction of the cell, almost an entire type of biomolecule it possesses. However, it also indicates that the ability to control PIP2 has a profound effect on the cell.
It also brings out yet another question. What else might control acclimatisation to the cold? Indeed, is there anything? The answers to these questions would tell us how such microbes perceive colder conditions, as well as what they require to live in them.
References
[1] I. Córcoles-Sáez, M. L. Hernández, J. M. Martínez-Rivas,
J. A. Prieto, F. Randez-Gil, Biochimica et Biophysica Acta, 2016, 1861, 213. http://dx.doi.org/10.1016/j.bbalip.2015.12.014.
[2] C. S. Ejsing, J. L. Sampaioa, V. Surendranatha, E. Duchoslavb, K. Ekroosc, R. W. Klemma, K. Simonsa, A. Shevchenko, Proceedings of the National Academy of Sciences, 2009, 106, 2136. http://dx.doi.org/10.1073/pnas.0811700106
[3] S. Furse, N. J. Brooks, A. M. Seddon, Rudiger Woscholski, R. H. Templer, E. W. Tate, P. R. J. Gaffney and O. Ces, Soft matter,2012, 8, 3090-3093. http://dx.doi.org/10.1039/c2sm07358g
]]>Control of the growth of single-celled organisms such as bacteria and fungi is a constant problem. We wish to encourage their growth in the industrial preparation of alcohols and anti-cancer agents [1] and in our digestive systems where they produce vitamin K2 [2]. On the other hand, we also want to rid ourselves of them when they infect us. Some infections, like tuberculosis, continue to be a problem. The clinical approach to curing tuberculosis in humans itself is famous because of the innovation of using several drugs with different modes of action, simultaneously.
Sadly, even this approach does not work for para-tuberculosis, an incurable condition similar to tuberculosis that affects ruminants, including cattle. The infection is a problem both for animal welfare and food safety reasons [3].
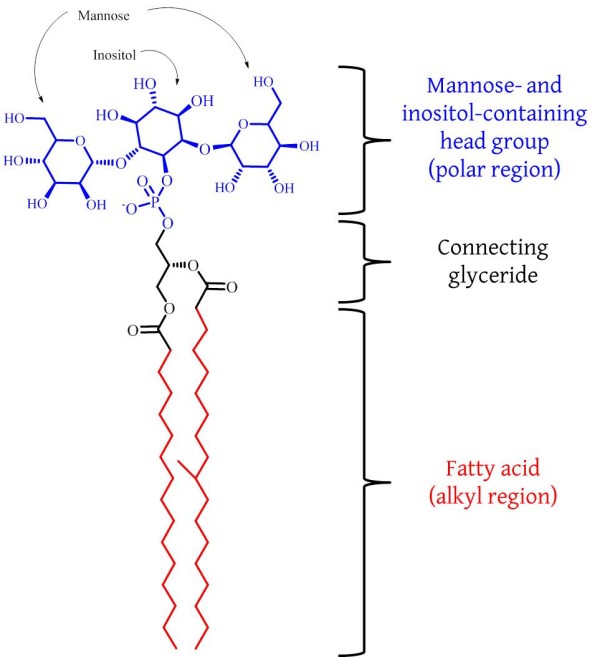
The incurable nature of para-tuberculosis raises a real problem for us to solve. Furthermore, it is an inter-disciplinary research problem: there are elements of microbiology/mycology, animal farming, toxicology, chemistry and maybe even food science. The work of such an unholy mixture of backgrounds can be informed and possibly even inspired by the work of a group of a Japanese-British-German collaboration, that was published recently. Hanashima et al. have shown that there is an endogenous protein in mammals that binds to a major component of the membrane of mycobacteria [4]. This component is called phosphatidylinositol mannoside (PIM) and is really a collection of several similar compounds [5, 6] of which a simpler example is shown in the Fig.
The protein is called ZG16p and was initially discovered in the pancreas of rats and later in the digestive system of humans [7]. There is therefore a protein that may be involved in the immune response to para-tuberculosis in all of us and may provide the basis for continued research into ways of tackling these kinds of infection without using anti-biotics. What if similar proteins could be engineered for tackling other diseases? This may be part of an adequate response to the increasing ineffectiveness of anti-biotics. And perversely, tuberculosis may once again provide the inspiration for our approach to tackling infectious disease.
References
[1] H. Ueda, H. Nakajima, Y. Hori, T. Fujita, M. Nishimura, T. Goto, M. Okuhara, The Journal of Antibiotics, 1994, 47, 301-310. DOI: 10.7164/antibiotics.47.301
[2] R. Bentley and R. Meganathan, Microbiology Reviews, 1982, 46, 241–280.
[3] N. Sung, M. T. Collins, Applied Environmental Microbiology, 2000, 66, 1334–1339.
[4] S. Hanashima, S. Gçtze, Y. Liu, A. Ikeda, K. Kojima-Aikawa, N. Taniguchi, D. Varûn Silva, T. Feizi, P. H. Seeberger, Y. Yamaguchi, ChemBioChem, 2015, 16, 1502 – 1511. DOI: 10.1002/cbic.201500103
[5] Y. S. Morita, J. H. Patterson, H. Billman-Jacobe, M. J. Mcconville, Biochemical Journal, 2004, 378, 589–597. DOI: 10.1042/BJ20031372
[6] G. D. Sprott, C. J. Dicaire, K. Gurnani, S. Sad, L. Krishnan, Infection and Immunity, 2004, 72, 5235–5246. DOI: 10.1128/IAI.72.9.5235–5246.2004
[7] H. Tateno, R. Yabe, T. Sato, A. Shibazaki, T. Shikanai, T. Gonoi, H. Narimatsu, J. Hirabayashi, Glycobiology, 2012, 22, 210–220. DOI:10.1093/glycob/cwr130
]]>
Humans, in common with most other animals and even bacteria, live and grow best at a moderate temperature with little change either up or down. On examining the physical behaviour of the lipids involved, we find a set of biomolecules exquisitely organised for our continued existence. There are lots of different sorts, and many organisms can adapt their lipid profile when conditions change a bit. For example, yeast can change the length of the carbon chains in its fatty acid residues to make its membranes less fluid. It can also introduce more unsaturated bonds into its lipids to make its membranes more fluid. However, the scope for this adaptation is limited both by what it can achieve (fluidity only goes so far) and that the genetic hardware for the different types of adaptation is not present all species. This raises the question of how we acquired the lipid profile we now have.
We could develop two hypotheses about the evolution of lipids in cells from the observations above. First, that the lipids we rely upon are difficult molecules that only really work well under quite particular conditions and our cells have exploited flexibility where it exists, but essentially, have obediently evolved around it. Second, that we have acquired a set of lipids that fits our purpose. That they have evolved alongside us, and the compromise that applies to all other aspects of evolution applies equally to lipids.
An observational comparison of the lipids in, say E. coli and Homo sapiens, might lean rather towards the first hypothesis. E. coli is made up principally of phosphatidyl ethanolamine (PE), with lesser amounts of phosphatidyl glycerol (PG), and cardiolipin (CL). Homo sapiens have all three of these lipids in plentiful supply in their cells. There are other lipid species too, but not in a way that makes one think that the two or three billions of years of evolution that separate humans and bacteria have reshaped the lipid fraction all that much.
The comparison between the lipids of E. coli and Homo sapiens is not really inconsistent with the second hypothesis, however—one might simply argue that humans and bacteria have similar requirements, such as a body temperature of 37 °C, and that is what shapes the lipids they have.
What we really need to conclude whether hypothesis one or two is correct is the existence of one or more lipid systems that demonstrably do not conform to the rules of ~37 °C, 0·9% salt and pH 7·4. We would have to see basically the same cells that we are familiar with but under considerably and obviously different conditions, and with different lipids.
This may be regarded as a good justification for searching for life on other planets. However, that approach has a fundamental flaw: there are terrestrial life forms that allow us to choose one of the hypotheses over the other. That is the domain of life known as Archaea. These are sometimes known by the slightly twee name of ‘extremophiles’ because they live in conditions that are different to our own preferred ones. The optimum conditions for growing Haloferax volcanii are 45 °C, 2·5 M NaCl and ~0·17 M Mg++ [1,2]. For comparison, this means it grows in conditions that are about 20°C warmer than we can comfortably live at, with 13× as much salt and very nearly 250× as much magnesium. There are others that seem less plausible still. Sulfolobus acidocaldarius have optimum growth conditions of 70-80 °C at a pH of about 2 [3].
Such conditions would hydrolyse the lipids that make up our cells. The ester groups that bond the fatty acid residues to the glycerol moiety* in would be hydrolysed, as would the phosphate moiety from its alkyl groups. Our cells would survive only for a few seconds. This raises the question of which lipids these extraordinary organisms have and what protection they offer to archaea that our own lipids do not confer on us.
First, the carbon chains are attached by ether, rather than ester, functional groups. These are much more resistant to acid- and heat-mediated hydrolysis. Furthermore, most of the lipids found in archaea are really ‘double’ lipids called bolamphiphiles. This means they have a head group at either end of a longer pair of chains. There are few or no unsaturated bonds. These features give a more rigid structure and may be the reason for other, counter-intuitive properties.
The higher temperatures that archaea grow at mean that deliberate efforts to confer fluidity on membranes do not seem to be as important. This means that the appearance of larger carbon rings than those observed in prokaryotic life forms: 5- and 6-membered instead of 3-membered, are remarkable. Furthermore, the hydrocarbon fraction of the lipids contains far more methyl groups than are typically observed in prokaryotes or eukaryotes. These also confer fluidity on the hydrocarbon fraction of the lipids.
There are several features that are very similar, however. The lipids in Haloferax volcanii also comprise analogues of PA, PG, PG phosphate and CL [4]. This is rather surprising because archaea are almost as distant a life from Homo sapiens as are E. coli. The evidence that these lipids appear in all three domains suggests that these lipids may be the oldest phospholipids. This is a tantalising clue as to what the common ancestor to all life on earth might have been like.
Despite this insight, there are still a number of questions that remain. Does the evidence that archaea have genomic strategies consistent with the ancestral life forms that gave rise to all current terrestrial life [5] extend to lipids, i.e. does it give us clues as to what the very first lipids were? Do archaeal cells shift their lipid profile through the cell cycle in the way that E. coli [6] and cells from Homo sapiens [7,8] do? At present, even the phase behaviour of archaeal lipids is not particularly clear. The third domain really has opened up a new line of research questions for both lipids and their role in vivo.
References and Notes
[1] T. Allers, S. Barak, S. Liddell, K. Wardell, M. Mevarech, Applied and Environmental Microbiology, 2010, 76, 1759–1769. DOI: 10.1128/AEM.02670-09.
[2] T. Allers, H. P. Ngo, M. Mevarech, R. G. Lloyd, Applied and Environmental Microbiology, 2004, 70, 943–953. DOI: 10.1128/AEM.70.2.943–953.2004
[3] L. Chen, K. Brügger, M. Skovgaard, P. Redder, Q. She, E. Torarinsson, B. Greve, M. Awayez, A. Zibat, H. P. Klenk, R. A. Garrett, Journal of Bacteriology, 2005, 187, 4992-4999. DOI: 10.1128/JB.187.14.4992–4999.2005
[4] G. D. Sprott, S. Larocque, N. Cadotte, C. J. Dicaire, M. McGee, J. R. Brisson, Biochimica et Biophysica Acta, 2003, 1633, 179–188. DOI: 10.1016/j.bbalip.2003.08.001
[5] M. Wang, L. S. Yafremava, D. Caetano-Anollés, J. E. Mittenthal, G. Caetano-Anollés, Genome Research, 2007, 17, 1572–1585. DOI: 10.1101/gr.6454307
[5] S. Furse, H. Wienk, R. Boelens, A. I. P. M. de Kroon, J. A. Killian, FEBS Letters, 2015, 589, 2726-2730. DOI: 10.1016/j.febslet.2015.07.043
[6] G. E. Atilla-Gokcumen, E. Muro, J. Relat-Goberna, S. Sasse, A. Bedigian, M. L. Coughlin, S. Garcia-Manyes, U. S. Eggert, Cell, 2014, 156, 428-439. DOI: 10.1016/j.cell.2013.12.015.
[7] C. V. Hague, A. D. Postle, G. S. Attard, M. K. Dymond, Faraday Discussions 2013 161, 481-497. DOI: 10.1039/c2fd20078c
*This is often called a ‘glyceride backbone’. I don’t know why because it’s more like the pelvis than anything. I think saying ‘glyceride backbone’ is an effort by science communicators to make this sort of molecule sound cool/accessible. I have no interest in being cool so I am opting for blind adherence to factual accuracy. I think that’s more daring.
]]>Several processes are essential for a cell to survive for long enough to proliferate. The release energy from chemical stores and the production of the machinery and structural components that make up the cell are well-understood, as is the disposal of waste. Smaller rubbish is generally easily dealt with, much of it diffuses out of the cell, requiring no further effort. But what about, for example, the material that is left after an infection?
Such debris constitutes large waste and is not material that can be expected to leave the cell by passive flow through the plasma membrane. This waste requires so-called Active Transport, a system that requires a co-ordinated effort to expel material through one or more membranes, including the plasma membrane. The steps are relatively straight forward, the material is rounded up and a membrane is wrapped around it forming a vesicle. The vesicle is taken to the plasma membrane, and the two membranes merge, expelling the contents of the vesicle into the extracellular medium.
So far, so intuitive. But how is this process, called exocytosis, controlled? What stops healthy or even essential material being lost this way, and how is all the cytotoxic* material expelled? These and other questions have been researched by cell biologists working on exocytosis. The control mechanism has received recent attention, with results showing that not only do lipids form the capsule that the waste is expelled by, but that lipids within that bilayer form part of the control mechanism. Recent work has focussed on how these control mechanisms operate, so that in disease states in which exocytosis is important, it may be manipulated accordingly. This has led to our being treated to two studies on the same subject, in the same journal, published at the same time.
The two papers offer a tantalising picture of what may occur through the whole control mechanism. Yamaga et al. report that removing a protein called PLC, that metabolises a signalling lipid, increases exocytosis [1]. In other words, the substrate for PLC increases exocytosis. The substrate is an old friend of this blog, PIP2.
Rogasevskaia & Coorssen [2] investigate another protein that is also known to act on PIP2. This protein is called PLD and is related to PLCs, but produces a different product, which is itself a signalling molecule. This molecule is called PA and is distinct from the lipid-like signalling molecule produced by PLC activity, called DAG.
The effects of PLD in producing PA are a modulation of the docking that occurs between the wrapped up waste (vesicle) and the plasma membrane (through which it must pass to be outside the cell) [2]. The effect of reducing PIP2 through PLC activity promoted dismantling of part of the cytoskeleton [1]. This makes it easier for incoming vesicles to meet with the plasma membrane and complete exocytosis.
These two observations invite several questions and observations. First, how do the PLC and PLD involved compete for the substrate? Having PIP2 around seems to help exocytosis to occur, but so does one of its products, DAG—or at least an absence of PIP2. Is PIP2 sacrificed for the good of the process, handing over the baton of waste management to younger lipids as it does so? It seems possible. What may also be required is a co-ordination of the amounts and activity of the PLD and PLC. It may also be necessary to control where they are, so that they PIP2 is sacrificed in the right way at the right moment.
What this work makes clear is that inositides and PIP2 in particular have a crucial role in cellular egestion as well as other signalling, such as proliferation and glucose metabolism. This underscores the question of how one lipid can have so many functions simultaneously and the control of all of them be retained.
References and Notes
*The prefix ‘cyto’ refers to cells. Thus, cytotoxic means toxic to cells, which may not be the same as toxic to organisms. Cytoskeleton is an internal structural support that contributes to the cell’s shape. Background on exocytosis and its antonym, endocytosis can be found here.
[1] M. Yamaga, D. M. Kielar-Grevstad, T. F. J. Martin, J. Biol. Chem., 2015, 290, 29010–29021. DOI 10.1074/jbc.M115.658328
[2] T. P. Rogasevskaia, J. R. Coorssen, J. Biol. Chem., 2015, 290, 28683–28696, Doi 10.1074/jbc.M115.681429
]]>
Diseases related to chronic lung damage kill about 25,000 people a year in Great Britain. That is about 40 per 100k of population. What this figure does not say is what it is like to live with the diseases, what might make them worse or cause it to start with, or what the underlying molecular changes are. Much of the damage involved in these conditions is due to long term exposures such as smoking and mineral dust.
Needless to say, physicians have ready advice for those suffering from the conditions. This advice has arisen from observations about the results of exposure to hot and cold weather, and pollution. Experience has also suggested that a particular geranium-scented gas is also a problem for sufferers of chronic lung conditions and can also damage healthy lungs, and thus by extension a low-level problem for the rest of us. It is heavily ironic that this gas should be dangerous, as the atoms that make it up are essential for life on Earth as we know it.
The gas is ozone. It is a molecule made of three atoms of oxygen. Oxygen molecules, that are essential to our continued existence, comprise just two atoms. Recently, it has begun to become clear exactly why this gas is so dangerous to lungs at a molecular level. Research carried out at Birkbeck College, University of London, using surface tension measurements has demonstrated that ozone interacts with a number of the molecular species resident in the lungs that are essential for its proper function. These include the molecular species that make up the bulk of the lipid fraction of lung surfactant [1, 2, 3]. Most recently, this research programme has produced evidence that the protein fraction of lung surfactant is also susceptible to low levels of ozone [4], also using surface tension measurements.
Surface tension is a measure of the physical properties of a lipid membrane or monolayer, and is useful because it affects the key role of the lungs, absorbing oxygen.
The protein studied is called Lung Surfactant Protein B (SP-B). It is made in the lungs and is located at the interface between the air and the fluid part of the lung. Like the lipids when they are exposed to ozone, there is a chemical reaction that causes an effective permanent change to the molecules affected. However, the effect of the ozone on lipids and the proteins is a bit different.
When the lipids are exposed to ozone, the surface tension of the surface the lipids form, goes down rather rapidly, before increasing to a point above where it normally is or should be for efficient lung function. However, this does not correlate with the effects observed after ozone exposure to humans [5]. The toxic effects of ozone are observed much later, quite unlike the effects of many toxic gasses such as cyanide or phosgene, that are essentially immediate. This raised the question of what else might be happening. It was certain that ozone was doing damaging things to the lipids, and that the lipids were essential for lung function, but the reason for the delay was not clear.
Interest therefore fell upon other components of the surfactant, to see what happened when they were exposed to ozone. Interestingly, the effect of ozone damage to SP-B causes an immediate increase in surface tension [5]. This is in the opposite direction to the decrease caused by the effect on lipids [2]. This delay does not last that long—perhaps half an hour in the experimental model systems—but is long enough to confound superficial observation of the effects of ozone on a mammal’s lungs.
The molecular approach to solving this problem not only answered an important question about lung function on exposure to ozone, it did so in a manner that could not have been achieved really any other way. Ideas about the relationship between surface tension, lung function and oxygenation had been established [6], but a physical molecular approach was required to get to the molecular heart of the problem. It represents a modern application of a scientific approach called reductionism: to break down a system into its component parts, and understand that system in terms of the behaviour of the individual parts and their relationships with each other.
Quite what the molecular relationship between ozonolised lipids and SP-B is in vivo is not at present clear, and thus may form part of a research question that has yet to be tackled.
References
[1] K. C. Thompson, A. R. Rennie, M. D. King, S. J. O. Hardman, C. O. M. Lucas, C. Pfrang, B. R. Hughes, A. V. Hughes
Langmuir, 2010, 6, 17295–17303. DOI: 10.1021/la1022714.
[2] K. C. Thompson, S. H. Jones, A. R. Rennie, M. D. King, A. D. Ward, B. R. Hughes, C. O. M. Lucas, R. A. Campbell, A. V. Hughes, Langmuir, 2013, 29, 4594−4602. DOI: 10.1021/la304312y.
[3] L. Q.ao, A. Ge, Y. Liang, S. Ye, J. Phys. Chem. B, 2015, Just Accepted Manuscript. DOI: 10.1021/acs.jpcb.5b08985.
[4] J. M. Hemming, B. R. Hughes, A. R. Rennie, S. Tomas, R. A. Campbell, A. V. Hughes, T. Arnold, S. W. Botchway, K. C. Thompson, Biochemistry, 2015, 54, 5185−5197. DOI: 10.1021/acs.biochem.5b00308.
[5] R. B. Devlin, K. E. Duncan, M. Jardim, M. T. Schmitt, A. G. Rappold, D. Diaz-Sanchez, Circulation, 2012. DOI: 10.1161/circulationaha.112.094359.
[6] M. Ikegami, T. E. Weaver, S. N. Grant, J. A. Whitsett, Am. J. Respir. Cell. Mol. Biol., 2009, 41, 433–439. DOI: 10.1165/rcmb.2008-0359OC.
]]>
It is a good time to be an alcoholic. The condition is far better understood than it once was, and more treatment than ever is available. The social understanding of the condition is beginning to shift away from it entirely dark and disreputable. In the western world at least, is more and more recognised as an addiction that needs treatment than a self-indulgence that should attract censure. It is less enigmatic, less sphynx-like. Furthermore, there is declining shame for taking the treatment for it. The damage alcoholism can do, and the damage it and its effects have, both physical and social, can be healed more easily than once they could.
This is tremendously promising for the furtherance of a social cause, and thus for improving a social problem. However, with respect to the medical problem, this impressive cultural change offers little help for the molecular or cellular effects of alcoholism. What is required for that is a scientific and data-driven approach. The first glimmers of a fresh angle to treating alcohol-related damage to humans has been published in the last month, through two (unconnected) papers.
Reichel et al. [1] used lipidomics techniques to investigate what impact alcoholism, and subsequent detoxification, had on the lipid profile of blood plasma. They found that the concentration of several lipids was higher in alcoholic individuals than in healthy controls. This included phosphatidylcholine and phosphatidylinositol. What they also found was that some lipids increased in concentration during detoxification. This included sphingomyelin, a lipid named after the enigmatic sphynx.
On its own, this is an interesting observation, from a sound study with a straightforward hypothesis. The observation gains momentum in the context of another study, by Yang and Subbaiah [2]. They found that an enzyme that clears fats from the bloodstream, particularly from the much maligned high-density lipoproteins (HDLs), is affected by the concentration of sphingomyelin.
This enzyme is called hepatic lipase. The name indicates that it has a close association with the liver (hepatocytes are liver cells) and that it catalyses a reaction that degrades lipids. What this enzyme does is hydrolyse both phospholipids and fats, releasing fatty acids. The activity of this enzyme correlates with a reduction in HDLs.
A reduction in HDLs might therefore be regarded as evidence of detoxification. This might in turn indicate that a patient has made it through the withdrawal stage and is making steps to recovery. However, HDLs have been identified as important in another medical arena.
HDLs are fat-carrying modules that are understood to be a factor in coronary heart disease. Where the levels of HDLs are low, the risk of heart disease is expected to be higher. A number of studies have investigated this link [3,4,5], some of them in an effort to distinguish between the lighter counterpart to HDLs, Low-density lipoproteins (LDLs) [5].
These two sets of studies are therefore an interesting comparison: on one hand, we have an increase in sphingomyelin that we would expect to lower HDLs, and on the other that lower HDLs increases heart disease. We might therefore conclude, on this basis, that for coronary heart disease at least, giving up alcohol is a bad idea.
Of course I am not about to tell you that. It would be naïve because the time interval is so short—we know that a short time where the HDL count is low on detoxification is not going to suddenly result in a coronary. However, what interests me about this, is that had this not been in the context of alcoholism and chronic illness, and the intuitive notion that a short term lowering of HDLs is probably not that bad, we might not have noticed. Had this relationship between sphingomyelin levels and the activity of hepatic lipase and detoxification been a purely cultural or purely molecular relationship, the same alarm bells may not have rung. We might just as easily conclude that ‘that sounds plausible’ and think no more of it. No enigma, no nagging doubt.
This indicates the value of complete evidence, and of testing things both ways. A passing relationship between two studies is exciting and may be relevant, and is good for bloggers, but human judgement is a crucial factor in making sense of it. And of that, we are all capable. Except, perhaps, before we have sobered up.
References
[1] M. Reichela, S. Hönig, G. Liebisch, A, Lüth, B, Kleuser, E, Gulbins, G. Schmitz, J. Kornhuber, Biochimica et Biophysica Acta, 2015, 1851, 1501-1510. DOI: 10.1016/j.bbalip.2015.08.005
[2] P. Yang, P. V. Subbaiah, Biochimica et Biophysica Acta, 2015, 1851, 1327–1336. DOI: 10.1016/j.bbalip.2015.07.003
[3] D. J. Rader, G. K. Hovingh, The Lancet, 2014, 384, 618.
[4] K. M. Ali, A. Wonnerth, K. Huber, J. Wojta, British Journal of Pharmacology, 2012, 167, 1177–1194. DOI:10.1111/j.1476-5381.2012.02081.x
[5] J. P. Després , I. Lemieux, G. R. Dagenaisa, B. Cantin, B. Lamarche, Atherosclerosis, 2000, 153, 263–272.
]]>
A social media storm ensued, and although the facts and independent evidence have only come to light recently—including a partial recording of the event, that shows the tone and reception of Sir Tim’s speech with what seems insightful clarity—the damage done to him has not been undone.
You can judge the evidence about what actually happened for yourself. I am not going to tell you what to think. So I have nothing to add to the reporting work done by Louise Mensch or Jonathan Foreman, or the opinions given by others, for or against, like Prof David Colquhoun and Prof Athene Donald.
What I am more interested in is what was behind what happened. Was Connie St Louis seeking to advance the cause of women in science? I think she probably wanted to, however ham-fistedly it came out. In fairness to her, the fact that what she wrote was retweeted 600 times was probably not something she planned maliciously or made happen. I imagine she planned for what she said to stand out, but I would also argue that is as far as it is reasonable to assume it went. What she said was calculated to get attention, which it did.
Her writing and comments since the affair suggest that she thinks what she did was right, in fighting for her views to be heard. The question it leaves me with is whether the cause of women in science, as the political phrase has it, is any better off now than it was.
The point that the cause of women in science is aiming at is, as I understand it, a meritocracy. It is what I, as someone who has gone on record in The Guardian as being a feminist, also seek. Almost every scientist I have ever met feels the same. I cannot speak for St Louis, and would not dare, but I have never heard she or any other feminist say that they seek a situation where women dominate over men. So, a system based on intellectual merit it is, then.
There is a good intellectual case for a meritocracy, or at least as close as it is practically possible to get to one. A meritocracy means that intellect is well used, that good brains are not wasted and thus do not go untrained or unused—or, at least not for their trifling associations. A merit-based system means we can focus on progress and creative invention. On forging understanding in scientific research. On making things better.
The best bit is that on paper, this intellectual utopia is not that far off. Women of my age have all sorts of choices that my grandparents’ generation did not. Women can choose whether and when to have children, whom they want to marry, what they want to study at school and university. They have female role models at school, in important positions and in the media. There was a female prime minister from my birth to virtually when I entered puberty. Furthermore, the law now ensures that they have the same pay as men. The issue I have heard complained about in person now, and one that is inconsistent between European countries is the parental leave system. It’s hardly the same barrier as once it was.
I can feel the hackles of radical or militant (depending on your perspective) feminists rising at my pointing out the statutory equal pay between men and women. The existence of the law on this point is a fact though, even if it is argued that it does not happen in practice. And it is argued that it does not happen in practice, but only with one piece of evidence. A pay survey by the Office of National Statistics (ONS).
The response to this ONS survey is in itself a fascinating case-study of how some non-scientists respond to evidence, and how scientists do. This also has a resonance in how social media handled the Tim Hunt affair—but more of that later. First, scientists regard the ONS study as data, that may or may not constitute evidence of something. Others regard it as proof of a particular conclusion. Scientists avoid terminology like ‘proof’ because it is an absolute, something that does not really exist in science. The reality of a situation tends to be rather more nuanced than a yes or a no.
Second, to a scientist, it is not clear what the limits of the ONS study are. For example, it is not clear what account has been taken of the difference in pay between black employees and white. There are plenty of people who will argue vociferously that blacks are still routinely paid less than whites. It is part of the argument about the existence of institutional racism.
What about ethnicity? Polish immigrants are still white, but are they paid the same as British ones in jobs in the UK? It is doubtful. Certainly traditionally, some ethnicities have been at a disadvantage in Britain. Just ask an Irish person who lived in London in the 60s and 70s. And what about age? Is someone of 40 paid the same as someone of 60 who has the equivalent experience? That is again not clear, nor is it clear how it affects these figures. One last one, and the slipperiest of the lot, is personal relationships. A simple example is: person A wants to impress person B. Person B has a spouse, child or protégé who wants a job. Does person A factor that into the equation about whether to give person C the job? What about in considering , or remunerate them handsomely to make the point? In other words, nepotism.
These are important questions that need to be answered about the ONS study. However, the same evidence-based logic applies, or should apply, to handling of news stories. Did all those hundreds of retweeters of what Connie St Louis wrote, check the story? Did they read about Tim Hunt’s record on promoting women in science, or whether he had employed women and how they had done, and what they thought? It seems doubtful. What is more likely is that they retweeted it because it confirmed their convictions; it was what they wanted to believe (and why they won’t be retweeting about this article, except in white-hot rage).
However such questions about the ONS data and the retweeters, are only questions. They do not necessarily kill the data or the original wording stone dead. Something else does that. What kills the ONS data stone dead for the purposes of proving gender pay divide beyond reasonable doubt, is the lack of specific evidence. If there were a systematic pay gap between men and women it should be straightforward to prove. There would be case study after case study that showed it. Furthermore, there are a large group of people who would love to prove it, as it would confirm their convictions. As far as I am aware, there is no such proof. This just does not square with the notion that women are paid less than men. There was a time when there were paid less. Back in the bad old days of the 1970s, it was very clear that women were being paid less because they were women. There is clear evidence of that. But those times have passed. The world has moved on. The crisis as once it was is largely, if not completely, over.
The same is apparently true of Tim Hunt’s career. This is despite his record, and the support he has received consistently from his wife, a senior broadcaster and several of his former, female, students. The evidence has not been weighed up and a balanced decision made.
That is not to say that it has all been achieved and that we should not worry about being complacent. Gender should not be a more important criterion than intellect for filling an academic position. However, if meritocratic principles are applied, there is no reason to expect that we will become complacent on this point. We do not need to be frightened that it will all fall apart.
The current competition for grant money, jobs and studentships in science makes this unlikely, at least in the short term. I can speak from experience on this. Recently I was involved in the selection process for a PhD studentship. There were 47 applicants from around the world. There were a considerable number of factors involved in telling candidates apart—it would have been near impossible to make any coherent distinction based on gender, if we had wanted to. Needless to say, there was no question of choosing on the basis of gender, and doing so would have felt very wrong.
That is a feeling I have a lot. Perhaps that is my flaw—it simply does not occur to me to be sexist. I am an intellectual, so why would any other criterion for making such a decision enter my head? I am convinced that I am like that, others are too. I am sure I am not unique in taking the view that intellect is paramount.
Does Connie St Louis take the same view? I hope so, but the evidence does not really square with that conclusion. She argues constantly and in what is described as a rambling way that ‘Women have had enough!’ (A piece in The Guardian on 23rd June 2015). That is not really the same as ‘All should be treated equally and equitably, based on intellect’, it is more akin to a mob shouting ‘Action Action’.
Perhaps I am being naïve. Perhaps she does agree with me but has translated her words into a sort of politicalese, in order to accelerate affecting the change she desires. It would be understandable. Many people take their causes into the political arena to effect a change and need to adapt to that environment. The evidence for this being a good way to change intellectual things is not universal though. In other words, politics is a blunt instrument. It is good at wider things in which it has an interest, like the economy. It is not a way of making nuanced changes to culture and fashion.
The real danger is that the use of politics to solve social problems has been tried before and with disastrous consequences. French revolutionaries, after they had killed off their aristocracy, started guillotining each other. Anyone who did not expound the most vociferous revolutionary views was regarded as a danger and was put to death. Robespeirre is a good example. It was a panicky response in a tense atmosphere and has proved far from stable (France was on its fifth republic less than 200 years after the revolution, where Britain has had a continuous monarchy since the interregnum). No one is expecting the Tim Hunt affair to turn into state-sponsored death, but ostracising someone from the scientific establishment is about the closest thing.
I am not really sure when feminism stopped being a poltico-social movement and started becoming a party political one, but there is no doubt that it has. It is not a move that fills me with hope, when evidence is mishandled, misreported and when lies take over from the truth. It is not helping the cause, it is taking it away from the place moderate feminists seek to arrive at.
Sexism as once it was, is dead and buried—and good riddance. So what sort of sexism is there to fight against now? The evidence is that in science women were employed 2:1 over men for first-run-on-the-ladder lectureship positions. But no one is going dare to support that view, despite the evidence of sexism. It looks worryingly like a meritocracy is not the desired outcome after all, but something else. Something more short-termist and probably egotistical. Feminism is needs a careful rethink on where it is going if it is not to be accused of being a lot of silly, head strong complainants who are wailing jealously for the sake of doing so.
]]>The question of where we have come from has fascinated—and frustrated—intellectuals of all sorts, for centuries. There have therefore been experiments in several learned disciplines to investigate this problem. Between them, they have produced lots of answers to the question. One such discipline is cell biology. The question in this context is how a newly born human baby, a viable multi-cellular organism, can form from just one cell. A human live birth at full term comprises about 716 billion cells. This means that cell division produces an average of 2.6 billion cells per day of the pregnancy. It is hard to imagine that—but the number itslef is not that important: all we need to know for these purposes is that it is a lot.
Unfortunately, there are analogous and far less happy examples of cellular growth at this speed. Cancers typically start with a mutation in a single cell, forming one that typically divides so rapidly and without a means to stop it, that it can overwhelm the system in which it lives. The system of controlling cell division, and thus the cell cycle, has got lost, been turned off or been inhibited out of use. Both systems containing cancers, and healthy ones that do not have been researched by cell biologists, with a view to determining how their cell cycles are controlled.
The work of Sir Paul Nurse and Sir Tim Hunt (described on the and in a BBC documentary established the way the cell cycle is controlled in eukaryotes. It is controlled by changes in gene expression, by changes in the concentration of proteins called cyclins.
I first heard about this in 2010, when I was writing my PhD thesis. This was years after the Nobel prize has been awarded in 2001 and therefore longer still after it had been published. This sat in my mind until about two years ago, when I was researching E. coli for a quite different purpose. I stumbled upon evidence that there are no analogues of cyclins in bacteria, and that gene expression does not change in bacteria during the cell cycle [1]. What interested me about this work was that the question of what was controlling the cell cycle.
I looked into other details of the system. For example, structural proteins are made and assemble into a sort of internal scaffold that supports the physical process of division. However, their concentration does not change through the cell cycle, so it is not reasonable to think they are at the centre of controlling the cycle either [2].
One possibility, I thought, was a fundamental of the process. In order for a cell to divide, it must grow longer (elongate) in order that the two cells produced are large enough to be viable. This means that the cell envelope—the system comprising the cell membrane—must increase in size by a factor of two before division. So, at the very least, the membrane limits the rate of cell division. I developed the hypothesis that it controls it, too.
The principal component of the membrane is the lipid fraction. There are proteins as well, some of which produce lipids, but ultimately most of the membrane’s area is lipids. This means that in order for the cell to divide, a big effort in making lipids is required. This is simplified in E. coli because it comprises only about 4 major lipid components. One of these, phosphatidic acid (PA), typically represents less than 1% of the total and is thus often ignored. Another component, phosphatidylethanolamine (PE) often represents more than 80% of the total and thus dominates. Of the two remaining, phosphatidyl glycerol (PG) is the principal substrate for making cardolipin (CL).
Furthermore, evidence about how these lipids are spread out through the membrane, has been researched and reported. There is evidence from studies where cells were dyed that CL is found at the ends (poles) of the cells. This meant that when cells were elongated, the percentage of CL would be lower. This is because the poles have stayed at the same size but the middle of the cell is longer. This offered the tantalising hypothesis that the cell might control its lipid profile, only producing CL when it made new poles, for example during division.
We therefore tested the hypothesis that the lipid profile of E. coli changes through the cell cycle. The next obvious question to answer, if such a study was to take place, was how this should be done. We elected to arrest the cells in two of the three periods of the cell cycle, the resting state and the state in which they are at their most elongated. The lipid profile was determined at each (See figure). The results were that the CL fraction fell in the way we expected, but also that the PG fraction increased a lot. In fact, there was a higher percentage of PG in the elongated cells than in the ones in the resting state. This suggested that the rate of its production was faster than that of the cell’s elongation [3].
Figure. The cell division cycle in the bacteriumE. coli MG1655. The resting state (B period) of the cell is the most familiar, and in cell division is the one that progresses to replication of the nucleic acid structures (nucleolus, C period) before full elongation in the D period, during which the cell undergoes physical division. The representation of the D period is the state of the cell when arrested by rifampicin: the cell has elongated fully, but the system is unable to move closer to division. Lipid profiling data from reference [3].
This fast production of PG is consistent with the considerable evidence that it is the principal substrate for the synthesis of CL in vivo. The last result was that the PA fraction also appears to fall when the cells elongate, suggesting that it too is found at the poles, with CL. These results led us to conclude that lipid synthesis is controlled as a function of the cell cycle, with synthesis of the different lipids turned on and off [3].
These results raise several questions. One is whether this type of controlled lipid production is the norm in bacteria. Work by Yao et al. [4], also published this month, suggests that another Gram-negative bacterium, Chlamydia trachomatis has a very different way of making its membrane than previously thought. The reduced genome of this organism had led scientists to the conclusion that it acquired lipids from its host cells. However, this recent work shows that C. trachomatis has an approach to lipid production that appears to be quite similar to that established for E. coli. This work provides compelling evidence that this bacterium can produce its own PG, CL and PE from PA, rather than acquiring its lipids from the host as previously thought. Not only does this insight inform us about the control this bacterium has over its membrane, but it suggests that lipid production may be similar across a variety of Gram-negative bacteria.
Another question that the work on the lipid profile of E. coli as a function of the cell cycle raises, is whether anything similar happens in quite different cell types, such as human cells. Atilla-Gokcumen et al. [5] and Hague et al. [6] who showed, by arresting HeLa cells at slightly different points of the cell cycle, that the lipid profile of the membrane of this cell type changes as a function of the cell cycle. Atilla-Gokcumen et al. went further still, showing that lipid distribution and the physical properties of the lipid mixtures also changes through the cell cycle [5].
It is not clear what impact the change to the lipid profile in HeLa cell membranes has on the control mechanism of the cell cycle in these cells. The control mechanism I mentioned towards the beginning, that involves cyclins, makes it too difficult to be clear about this mechanism from the current data alone. However, it raises an interesting possible comparison. On one hand, there are eukaryotes who have developed from prokaryotes, and comprise a protein/gene-expression based cell cycle control system but in at least one cell type have significant change in lipid profile through the cycle. On the other, prokaryotes do not have the same protein-control system for the cell cycle as eukaryotes do, but also change their lipid composition through the cell cycle. Is change to the lipid profile through the cell cycle thus a fundamental part of cell division, whether or not it is the controlling mechanism?
There is much work to do to establish this, and so the story stops here for now. However, it may well go further forward in the next few years as it is conceivable that answering this question could support other human endeavour. This gives a social argument for funding. For those interested in the social and human angles of scientific research, the applications of understanding how the cell cycle of bacteria is controlled are at least two-fold. In cases where we might wish to stop them growing (in infection control) or to promote it (in industrial fermentation), what limits the cell cycle is important, as it provides both what can be exploited to arrest growth, and what must be supported to promote it.
References
[1] S. J. R. Arends, D. S. Weiss, Journal of Bacteriology, 2004, 186, 880-884. DOI: 10.1128/JB.186.3.880-884.2004
[2] S. Rueda, M. Vicente, J. Mingorance, Journal of Bacteriology, 2003, 185, 3344-3351. DOI: 10.1128/JB.185.11.3344-3351.2003
[3] S. Furse, H. Wienk, R. Boelens, A. I. P. M. de Kroon, J. A. Killian, FEBS Lett., 2015. DOI: 10.1016/j.febslet.2015.07.043
[4] J. Yao, P. T. Cherian, M. W. Frank, C. O. Rock, Journal of Biological Chemistry, 2015, 290, 18874 –18888. DOI: 10.1074/jbc.M115.657148
[5] G. E. Atilla-Gokcumen, E. Muro, J. Relat-Goberna, S. Sasse, A. Bedigian, M. L. Coughlin, S. Garcia-Manyes, U. S. Eggert, Cell, 2014, http://dx.doi.org/10.1016/j.cell.2013.12.015.
[6] C. V. Hague, A. D. Postle, G. S. Attard, M. K. Dymond, Faraday Discussions 2013 161, 481-497. DOI: http://dx.doi.org/10.1039/c2fd20078c
Conflict of interest statement: I am the first author of reference [3].
]]>Tweet
‘When will we beat cancer?’ is a question asked of a lot of scientists who work in cancer research. It was a question I was asked intermittently, when friends-of-friends heard that I was (nominally) involved in cancer research as a PhD student. To begin with, I took a sagacious approach, explaining that cancer is really several conditions, which can differ markedly from one another. I also mentioned what I thought to be fun facts, that the disease can change during its course within individuals and so beating it was a difficult thing to pin down. This was rather an effort and often made people look at their glass whilst I was speaking. So after a while, I replied with a question ‘Have we beaten infection?’
This manages to hint that we probably never will beat cancer entirely, but we can find out a lot about it and exercise a good deal of control over it. The battle with infection has seen diseases that were once widespread, like polio and tuberculosis, largely disappear (anti-vaccination campaigning notwithstanding). Many of these disappearances were unimaginable at the turn of the 19th century. The fight against infections such as these has also affected us in more subtle ways. Not only do we live longer, but we understand how to manage and prevent non-life threatening infection, such as a lot of food poisoning. This has led to had a considerable effect on how we eat—refrigeration is a good example.
So, if tackling cancer is like the fight against infection, it too will affect us in ways we cannot imagine at present. This makes broader examples rather interesting—not only do they contribute to the desire to meet challenge of controlling cancer, but they offer tantalising possibilities of other things. One recent development, that may have the potential to influence other things, is an anti-cancer therapy that interferes with a whole metabolic pathway. This type of treatment affects a number of biochemical reactions and therefore the breadth of its effect is wide.
At first glance, this sort of approach seems a bit unrealistic, even cowboyish. Disturbing a whole molecular pathway, rather than a clear, narrow target has many potential dangers. There is a lot going on and it is easy to imagine that some of those effects may be undesirable or even harmful. It may even be that it creates a situation in which the cure is worse than the illness. Despite such pitfalls, recent evidence shows that it may be possible to use a broad tool for just such a purpose. This involves a known drug called Tamoxifen.
A recent study by Morad et al. showed that Tamoxifen can increase the death of cancer cells when given as part of a set of drugs [1, 2]. It does this by blocking the production of fully-fledged sphingolipids from ceramide, and by blocking the destruction of ceramide. This double-effect of ceramide not being broken up, nor made into other lipids, means that it can accumulate in cells. The effect of this result has a firm background: the presence of ceramide has been known for at least a decade to lead to programmed cell death (apoptosis) in cancer cells.
Despite this clear result, it is not yet clear how the drug achieves this [3]. One suggestion is that it has a few concurrent effects that have yet to be assessed together. The effects of Tamoxifen are still being discovered: another recent study, Khadka et al., details the effect Tamoxifen has on the physical behaviour of membranes. This study indicates that the barrier properties of membranes appear to change in the presence of tamoxifen [4]; cell membranes are weakened, causing greater leakage. This means the cell cannot maintain its internal environment and almost literally falls to pieces.
Thus, where tamoxifen can be targeted to cancer cells, it has real potential as a tumour-killing drug. This adds to its use as an established anti-cancer treatment; it is currently used in treating breast cancer, in which it works by modulating the activity of oestrogen receptors in the system [5]. The discovery of a second use for tamoxifen is also convenient because its clinical use means that it is already an accepted and available drug.
It is appealing to speculate about the broader impact of this understanding. Perhaps it will be possible to have a multi-drug broad-spectrum tablet or injection that can tackle a variety of cancers at once. This might mean that some of the most dangerous cancers, such as those at later stages when the cancer has spread (metastasis) become treatable. This may influence the desire for cancer screening, either reducing it (because cancers are easier to treat) modifying it (some cancers do not need to be screened for, others will be screened for more carefully) or extending it (catch it all early and minimise drug administration).
Any of these might be expected to have a knock-on effect on screening technology and its secondary uses. For example, if cancers that Tamoxifen may not be used for are screened for more carefully, for example in a given organ or organ system, our understanding of that organ or organ system may increase. Lung disease may be understood better as a result and thus how we condition air and what we consider ‘good air’ may therefore change. Screening for cancers in reproductive systems may be understood better, giving an insight into infertility or contraception.
These possibilities are of course guesswork at present. Readers, as always, are encouraged to comment, but particularly on this occasion: what might the fringe or unexpected benefits of treating cancer more fully, be?
References
[1] S. A. F. Morad, S. F. Tan, D. J. Feith, M. Kester, D. F. Claxton, T. P. Loughran Jr., Brian M. Barthd, Todd E. Fox, Myles C. Cabot, Biochimica et Biophysica Acta, 2015, 1851, 919–928.
[2] S. A. F. Morad, Myles C. Cabot, Biochimica et Biophysica Acta, 2015, 1851, 1134–1145.
[3] T. A. Taha, T. D. Mullen, L. M. Obeid, Biochimica et Biophysica Acta, 2006, 1758, 2027–2036.
[4] N. K. Khadka, X. Cheng, C. S. Ho, J. Katsaras, J. Pan, Biophysical Journal, 2015, 108, 2492–2501.
[5] V. C. Jordan, British Journal of Pharmacology, 1993, 110, 507–517.
]]>Tweet
In 1945, Fritz Albert Lipmann made a discovery that would earn him a share in the 1953 Nobel Prize for physiology or medicine. He (co-)discovered of Acetyl Co-enzyme A. Little did he know that seventy years after that discovery, we would find enzymes that use this co-factor in places as obscure as the genitals of an insect [1]. Less still did he expect bloggers to snigger inwardly whilst they wrote about it.
In fairness to him, it would be strange if he had. He was concerned with understanding how metabolism works, specifically, how energy is released from the chemical stores we rely upon. These are the mechanisms we have for both normal metabolism (being alive) and increased metabolism (exercise). It is fashionable at present for both sugars and fats, and the foods that contain them, to get a howlingly bad press in most of the increasingly obese western world. But how much do we know about the journey from 100 g of chips to a 4 kilometre run? Or to a bumblebee’s bollocks?
The energetic route is probably the better known bit. Chips, sliced potato deep-fried in vegetable oil, have about 180 calories per 100 g. That means, for the average 25 year old who weighs 70 Kg, 100 g of chips is enough to power a jog of just over 20 min. (I used the British Heart Foundation exercise calorie counter for these calculations, but there are others.)
Part of the gap between the eating and putting one leg in front of the other is called beta-oxidation and the overall chemistry of it is that it replaces the hydrogen on the carbons of the fats, with oxygen. It is the beginning of the process that turns the fat into carbon dioxide.
In beta-oxidation, the fatty acids bonded to coenzyme A are snipped into two-carbon units. The four-step process that does this leaves acetyl Co-A and another Co-A with the rest of the fatty acid on it each time. The snipping process is then repeated seven or eight times until the fatty acid has been used up—most fatty acids have 16 or 18 carbons. When used just for energy, these Acetyl Co-As are fed into the Electron Transport Chain, the final step of primary metabolism in which ATP, the ‘cash’ of energy provision in vivo is made. The citric acid cycle part of it is the one that releases the CO2, the organic product of the metabolism of fats.
Acetyl Co-A is more like the gold bullion of the internal biological energy market. It is not necessarily used for money, it can be used for other things. It can feed into secondary metabolism.
Acetyl Co-A is tremendously useful for secondary metabolism as the small 2-carbon units are inherently flexible building blocks. They can be used to make larger building blocks, such as isopentenyl pyrophosphate, IPP. This is where the preparation of sophisticated signalling molecules may enter from stage left, carried in a bee’s knickers. Specifically, 2,3-dihydrofarnesol. This is the chemical name of a molecule made from IPP that sadly gives nothing away about what this molecule does, or where it comes from.
It comes from the labial gland of Bombus terrestris. These are bumble bees, and it is the males that have said glands. 2,3-dihydrofarnesol is a sex hormone that is used for ‘pre-mating communication’. (This sounded like a euphemism to me, but it is not, it is really a way of referring to a part of a longer process that is affected by all sorts of factors, such as the timing of light and dark [2].). It is part of the system that leads to successful reproduction of Bombus terrestris, which one hopes maintains the population of pollinators and thus the reproduction of plants, by far the biggest producers of fats—and the only producers of potatoes—around.
This great pathway, that takes us from fat, to bees, and ultimately back to fat is part of a system with all sorts of other routes and cycles too long to go into here. So, the simple message is this: next time you look at a plate of chips, just think ‘A bee did that’.
References
[1] J. Brabcová, Z. Demianová, J. Kindl, I. Pichová, I. Valterova, M. Zarevucka, ChemBioChem, 2015, 16, 1047-1051. DOI: 10.1002/cbic.201402591.
[2] Y. J. Kwon, R. Amin S. J. Suh, Apidologie, 2006, 37, 679-686. DOI: 10.1051/apido:2006047
Further Reading
Biochemistry, Lubert Stryer. Various editions, Freeman Press.
Great for University-level understanding of the metabolism of fats and other biomolecules
Secondary metabolism, J. Mann. Various editions, Clarendon Press/OUP.
An excellent University- and professional-level reference on the biological preparation of compounds such as 2,3-dihydrofarnesol.
Tweet
There were some political protests this week. One in London and one in Cardiff. They were campaigning against the Conservative policy of austerity. The damnedest thing of all was that the poor sods were a day too late. A little glitch in their event management meant they did it they day after the election instead of the day before. It could happen to anyone.
I am being facetious, obviously. It was of course a response to the result of the general election, rather than a way of drumming up voters to back their cause. It has happened a few times before, after Conservative governments have been elected. There is therefore a history, or perhaps a tradition, of British left-wing activists doing that. Oddly, right wing ones have not adopted the same tactics. Conservative election wins are invariably met with scorn protest where Labour ones are not.
Perhaps the answer to this is that the governments are elected for different reasons. There is the feeling that Labour governments are elected mainly because Conservative ones are unpopular, rather than because the Labour prospect is itself popular. Labour have a dedicated following of course, which is probably smaller than the tory one, at least if the number of years of conservative government since WW2 is compared to that of Labour. It is about twice as much.
I was surprised when I first heard that statistic. Perhaps it is all the more surprising when we consider that voting conservative is largely socially unacceptable. Or, at least tories are shy, according to Lewis Barber in the Independent. This shiness might also be why so many of the pollsters got it wrong and why the BBC’s poll that predicted the conservative party would be the largest was published so nervously on election night.
It would be easy for right-wingers to blame a left-biased media for this sort of thing. Realistically, they had no direct role, and I think we can ignore evidenceless media conspiracies as the mechanism behind voting practices. The same applies to the protests: they were not media-generated. By far the most likely explanation for those was that a group of lefties did not get what they felt entitled to at a general election, and rocked up in a couple of big cities (and not even the most Labourite ones) to say so.
This analysis has them as childish, and worse still, undemocratic. It suggests they should vote for what they want and then move on whether or not they get it, at least in the short term after the election. That is probably what Conservative voters do, but as we know they have different behavioural response to electoral disappointment. The only Conservative-type protest in Britain that I can think of is the countryside march. That was in the middle of a parliament, partly a response to the Hunting Bill. Despite being huge and non-violent, it was not listened by the government of the day any more than any of the others were, so was probably as just as ineffectual. Still, we retain the view and practice that people having their voice non-violently is part of democracy and no one in the UK is about to stop it.
No one needs to tell the Scottish nationalists this. They did not get what they wanted in the referendum on Scottish Independence, but there was no crying or stamping of feet. They worked out what to do and then did it. Now, 56/59 Scottish seats in the house of commons are held by the SNP, who got something like 50% of the vote in that Kingdom, overturning several considerable Labour majorities in the process. They now have a strong ‘Scottish voice’ and probably the political will and power to get their way. Their respect for democracy has been exemplary throughout.
The SNP has also been campaigning for independence for 80 years. In context, that’s about twenty years longer than the National Health Service, an important socialist achievement, has been in existence. We still have a National Health Service, and have even added to its remit the task of being something for everyone to complain about. We also have the Hunting Act and the Union remains intact. So, who has got the best political tactics?
]]>Tweet
The broadest definition of lipids includes fats and cholesterol as well as biological amphiphiles. This means that the study of obesity is often regarded as part of the study of lipids. In fact, the study of obesity is a good broad definition of the clinical discipline of lipidology. The principal stuff of obesity is of course fat, referred to formally as triglycerides.
The structure of triglycerides give very little of their peripheral importance away: the social, emotional and even political meaning these molecules have is not at all evident from the molecules as we see them on the page. In fact, despite considerable research into human metabolism over the last hundred or more years, there has been very little impact on obesity. If anything, Europeans and North Americans are fatter now than they were a century and a half ago. This is despite the increase in our understanding of fats and the dangers they pose. This ‘obesity crisis’, if a crisis can happen as slowly as someone over-eating, also correlates with the popularisation of dieting.
In 1862, the undertaker by Royal appointment to the House of Hannover, a William Banting, published a ‘Letter on Corpulence: Addressed to the Public’. The document is still widely available, and makes interesting reading for social historians as well as lipidologists, and probably those interested in the psychology surrounding losing or gaining weight. It is about what became known as Banting, a form of dieting.
A look beyond the Victorian verbosity and veneration brings out familiar themes. He describes the perceived difficulties of corpulence—of being unfit, of having an awkward bodily shape, of lacking energy. He also fashions a superficially believable excuse for not increasing his calorific expenditure (taking more exercise), and hints at many of the ignorances of the time, including that fatness is age-related and that gout and obesity are linked.
Two particular things about this Letter stood out for me. First, that this appears to be an example of the belief that weight loss can, or should, or should only be attempted purely through dietary change. Second, that this is the first low carbohydrate diet, and forms the basis for all those that have been constructed in that vain since.
The idea that losing weight can be achieved through changes to diet alone is not necessarily wrong—no one who understood anything about human metabolism would tell you that it were—but equally there is no doubt that it is not the full story. The mass of triglycerides (amount of fat) in our adipose tissue can regarded rather like the amount of money in a bank account. If we spend more (take more exercise), the balance falls (we get thinner). If we earn more than we spend (eat more calories that are used), the balance goes up (our adipose tissue stores more fat). So, rather like running a current account, the balance between what is spent and what is earned is the key to changing the size of fat reserves. It is not just about what is earned.
Banting’s unwitting begattation of the ‘low carbohydrate diet’ is a bit harder to analyse. Certainly, he consumes fewer calories from starchy foods on his weight loss diet, to a level that he eats fewer than he needs and thus uses up his reserves. However, the diet also represents a considerable reduction in the intake of fat, and not necessarily as low a carbohydrate intake as one might imagine. He avoided pastry and fattier meats but also drank far more alcohol than is recommended today. The emergence of ‘low carb’ diets from this one is therefore a bit of a mystery. It was probably not widely understood in the 1860s was that fat contains more than twice as many calories as either protein or carbohydrate, gramme-for-gramme. Could it be that this is still poorly understood today?
Food is a complicated mixture of molecular species, and indeed the composition of foods is too complicated even to be at the centre of food science. Biochemical studies mean we now know that our omnivorous digestive systems and evolved metabolic machinery are well able to deal with a variety of molecular species. This means that we can tolerate a great variety in our diet. Our bodies can make fats out of both carbohydrate and protein, but cannot make either protein or carbohydrate out of fats. We can convert some protein monomers (amino acids) into others, and thus make up for a certain deficiency of amino acids in our diet, but by no means all—there are at least eight amino acids we must have in our dietary intake, the best sources of which are meats.
Human bodies are not really able to make carbohydrates at all–at least not ones for storing energy. Though we can get energy from fats, we rely upon carbohydrate absolutely. In fact, the basis of all terrestrial metabolism is the oxidation of glucose to carbon dioxide. Thus any organism that respites requires a supply of glucose. This means that although there is scope for a good deal of variety, the evidence is clear: there are some things we cannot expect to change.
However, the fact remains: no matter how clear the data, how good the science, or how thoroughly the fad diets and the bullshit of phrases like ‘low carbs’ are debunked, obesity remains. The science is not enough on its own, to stop the crisis. This suggests to me that the study of obesity is the study of the problem, more than the solution. Where the solution will come from is harder to say—but a hundred and fifty years of Banting has not yet given the answer.
References and Further Reading
Letter on Corpulence: Addressed to the Public, William Banting. (A simple google search will bring this up though links here, here and here work at the time of writing)
The Fats of Life, Caroline M. Pond. A dry and now somewhat dated but easy read on fats. More recent research includes evidence that illuminates our understanding of the genes involved in the metabolism of fats, see here.
]]>No one likes being on the receiving end of pedantry. Typically, the only thing implementing that feedback protects one from is other pedants, and so a pedantic response is often nothing other than unattractive. Despite that, Fry’s arguments did not sit that well for me, and I have finally decided to let the tip off my tongue trip of the top of my palate to say so. Or at least write it on my iPad.
It is certainly true that the point of language is not to get grammar and punctuation spot on. No one said of Jane Austen that she was a master of the semi-colon, that her use of the apostrophe was unimpeachable. In a sense, at the distance we now are from her, it does not matter whether she knew how to use punctuation marks or not. We rather assume that she did, because whoever wrote Pride and Prejudice was clearly very intelligent and so was almost certainly capable of grasping the difference between possessive and diminutive, and that neither was necessarily a plural. It does not matter to us whether or not she knew this, nor showed whether or not she knew it. The rule that gives the full meaning to girls’ bags, remains.
In fact, any deviation from this simple rule is capable of moving the meaning to something that either renders the sentence incomprehensible or for it to sound as though it means something other than what was intended. There are of course countless instances where it probably does not matter to the meaning the writer wishes to convey, at least to those sufficiently competent in the language. A common argument against this lugubrious language use is that it is at best lazy not to polish, and at worst, inconsistent and difficult for non-native speakers.
As someone who writes a lot about science, I typically make an effort to get it all right. This partly comes from the experience that so many people see my copy before it gets published that someone will correct me if it is wrong. Either that, or they will correct it to what they think it should be, which results in a poor piece of writing (cf. death by committee).
In the last two years, I have been living in a European country where English is the lingua franca and so I have learnt about attitudes within and towards other languages. This has shifted my perspective and allowed me to articulate my view about why arguments that ‘it doesn’t matter if your grammar and punctuation are wrong, no one cares’ do not sit well.
It is because language relies upon rules to be understood. I agree with Michael Rosen that English is formulated more from patterns than from rules, unlike many European languages in which any deviation from the set order results in confusion, and where sentences therefore have an unambiguous meaning. It is hard to apply that to English—the use of tone means that we can make the same sentence mean five different things. That makes written English by definition a bit ambiguous. Widespread use of a common rule book is therefore impossible—English is intractable.
However, I question whether that is a strong enough argument for tacit acceptance of not following the pattern, especially where it leads to ambiguity. Another way of thinking of this is that although patterns can be broken or remoulded in a way that rules cannot, the fact that someone clever does it, does not really give all of us licence to do so.
I broke the rule about not starting a sentence with a conjunction or a preposition twice in the first paragraph of this article, and I probably got away with it. The way I constructed the paragraph meant that I could probably get away with it. Was it really worth breaking the rule, though? Did I use it in an elegant enough way to justify alienating pedants and confusing those who are doing battle to make sense of this rule-less tongue?
Probably not. And that is really the point, if it does not have a purpose, is it really fair on the reader?
]]>Tweet
The Origins of Life on Earth is a topic that has an impressive resonance. It comes from an innate human desire to belong: almost everyone wonders where they came from. Anyone who has been to a Natural History Museum, and many who have not, wonder what the world must have been like when the dinosaurs were around. Almost everyone fortunate enough to have had a decent science teacher will have been put in the way of wondering what life was like before any kind of multi-celled organism. Geologists and palaeontologists have pieced together evidence from their respective fields to give a time line of the Earth’s history (Figure 1).

Figure 1. A time line of the geological and palaeontological events that we understand of the Earth’s history. This figure courtesy of Astrobiology Magazine, original credit: Dan Brennan/UW-Madison News Graphic
In the 1950s, two biological chemists took this curiosity a step further. Stanley Miller and Harold Urey tested the understanding of the early Earth by recreating the molecular components and conditions of the period, in the lab. They put ammonia, water, methane and hydrogen in a flask and simulated the conditions of an electrical storm (one with lightning) for a week [1, 2] (Figure 2). What they found amazed them. One thing was amino acids—the monomers that comprise proteins, the machinery of cells. The second, a bit further downstream, was evidence for ribose sugars. These are the precursors for nucleic acids, the basic units of the blueprints of life, RNA and DNA. At a stroke, there was evidence for prebiotic synthesis of two of the principal molecular species of terrestrial living systems.

Figure 2. The equipment set-up used by Miller and Urey to explore the chemistry of the early Earth’s atmosphere. It was with this that they showed that amino acids and perhaps riboses could form essentially spontaneously under these conditions. Diagram courtesy of Wiki commons, drawing credit here.
A subtle conclusion to this striking result is that the chemistry that drove the production of some of the basic molecular units of life is so simple that in terrestrial terms, it happens spontaneously. This provides an attractive explanation for the how the first batches of two major sets of biomolecules were made. It is not clear whether these spontaneous syntheses happened in rapid succession, as the Miller-Urey experiments suggest, or whether several molecular contributions and variations occurred over years or even millennia. What is clear is that stocks of these molecules could have built up well before they became part of living organisms—there was no ‘Frankenstein moment’ when life was suddenly conferred to a pool of chemicals on the Earth.
It is not a huge step of imagination from the riboses and simple amino acids we understand may have existed in this period, to the blueprints and machinery of the cells of which we too are made. What is missing from these experiments is the emergence of the boundary species, the molecular component(s) that separated the cell from its surroundings. What we have yet to pin down is the origin of lipids.
The emergence of lipids has a particular importance in the origins of life. Without such molecular species, it is not possible to construct even the most basic form of cell as we know it. The simplest requirement of a cell, alongside it being able to replicate, is that it can manage its internal environment. In fact, if it cannot control its internal environment, in practice, it cannot reproduce. An indispensable part of this is the barrier between the inside and the outside—in our cells, the lipid bilayer. Thus, lipids are required in order for cells as we know them to exist.
This need to explain the preparation of the earliest lipids presents several immediate problems in explaining how life as we know it got going. The theory and evidence we have is quite convincing from single cells up to mammals (any decent biology text book above a certain level will explain this lucidly), and the Miller-Urey experiments [1,2] give a significant insight into where amino acids and perhaps nucleic acid polymers came from. Work over the last decade shows that the genetics behind early cells is becoming clearer [3,4], indicating that some of the replication machinery of early cells is being better understood. But how do we get from the early Earth’s atmosphere and weather, to molecules that can spontaneously self-assemble into a primitive membrane that allows cells to divide? Moreover, how can this happen as spontaneously as amino acids form when lightning strikes the ammonia-methane-water-hydrogen cocktail?
A recent paper by Patel et al. begins to provide a possible answer [5]. They show that compounds such as prussic acid (hydrogen cyanide), in the presence of copper ions and hydrogen sulfide (also known as hydrogen sulphide, understood to have been released by volcanic activity early on in the Earth’s existence), can form precursors for nucleic acids, amino acids and, importantly, lipids.
The crucial part of the spontaneous formation of lipids is how polymers of methane the order of 16 or 18 carbon atoms formed, with oxidation probably only at one end, and how enough of these formed in close enough physical and chronological proximity to assemble into a bilayer. Sadly, this has yet to be explained; a Miller-Urey type experiment that results in fatty acids or anything similar, has yet to be reported*.
It is an experiment that many lipid chemists and lipid biologists would give a great deal to stumble upon. What I predict as a more likely progress to the answer is a meeting in the middle. Several experiments have shown that self-assemblies of simple fatty acids are sufficient to provide a boundary for RNA replication [4]. Such experiments have the potential to inform us about some of the limits of what is required for this to take place, e.g. What the shortest length of fatty acid is required for such a boundary, or whether or not fatty acids with oxygen or nitrogen functionality in the middle or at both termini (such as in archaea) are more energetically favoured. At the other end, the design of compartmentalised systems in which conditions such as pH may be controlled [6] tells us the minimum requirements for basic homeostasis. The (proto-)fatty acids that fall into both categories, ones that are easily made spontaneously and ones that can make a boundary, are prime candidates for being called the first lipids.
References
*I hardly need tell you that if I had a shed, I would be doing this sort of experiment in it.
[1] S. L. Miller, H. C. Urey, Science, 1953, 117, 528–9. DOI:10.1126/science.117.3046.528.
[2] S. L. Miller, H. C. Urey, Science, 1959, 130, 245–51. DOI:10.1126/science.130.3370.245.
[3] D. G. Gibson, J. I. Glass, C. Lartigue, V. N. Noskov, R. Y. Chuang, M. A. Algire, G. A. Benders, M. G. Montague, L. Ma, M. M. Moodie, C. Merryman, S. Vashee, R. Krishnakumar, N. Assad-Garcia, C. Andrews-Pfannkoch, Evgeniya A. Denisova, L. Young, Z. Q. Qi, T. H. Segall-Shapiro, C. H. Calvey, P. P. Parmar, C. A. Hutchison, Hamilton O. Smith, J. Craig Venter, Science, 2010, 329, 52-56. DOI: 10.1126/science.1190719
[4] J. C. Blain and J. W. Szostak, Annual Reviews in Biochemistry, 2014, 83, 615–40. DOI: 10.1146/annurev-biochem-080411-124036
[5] Bhavesh H. Patel, Claudia Percivalle, Dougal J. Ritson, Colm D. Duffy and John D. Sutherland, Nature Chemistry, 2015, 7, 301-307. DOI: 10.1038/NCHEM.2202
[6] D. Miller, P. J. Booth, J. M. Seddon, R.H. Templer, R. V. Law, R. Woscholski, O. Ces, L.M. C. Barter, Journal of the Royal Society, Interface, 2013, 10, 20130496. DOI 10.1098/rsif.2013.0496
Tweet
If I told you that a lack of lipids were responsible for causing ageing in yeast but too many is associated with insulin resistance in humans, and that although lipids dominate the membrane of the cell envelope in E. coli, they may not be essential, and if I added that lipids could be used to make fluorescent molecules that could be used to probe cellular systems, can be a raw material for making secondary messengers and also be a sort of chemical exchanger to repair other molecules, what would you say?
You might tell me, gently, that you have read The Lipid Chronicles before and so of course I am going to tell you that lipids are the renaissance–how on Earth would anyone be able to write 50 blog posts about a subject that did not have a bit of sparkle?
And you would be right, but perhaps in a crook. All of these things are not the responsibility of lipids, they are the responsibility of one lipid. Phosphatidylethanolamine, PE (structure shown in the figure).
Figure. The structure of phosphatidyl ethanolamine (PE). The head group and glyceryl moiety are shown in red and the lipophilic part is shown in blue. The NH3+ is the primary amine group, that exists in this protonated form at physiological pH.
No one would have expected PE to be a renaissance man. It is rather ordinary in many ways, old and a bulk lipid to boot. It makes up >70% of the phospholipid fraction of E. coli and does not really form membranes in its pure form. But in my opinion, therein lies much of the scope for the variety: what we see is that small changes can shift the behaviour of this lipid over the line in one direction or another, showing lots of different guises. This multi-guise idea is strengthened by another contributor, the nucleophilic primary amine group –NH2, that is at the top of the head group. This functionality is the principle difference between PE and phosphatidylcholine (PC) in practice, as well as being a precursor for it. This gives PE a distinct ‘feel’ with respect to other lipids, especially other bulk lipids with a similar structure. This uniqueness gives rise to the ready identification of PE as responsible for its actions.
There is evidence that a lack of PE appears to be responsible for premature ageing in yeast [1]. This comes from a study by Rockenfeller et al. in which the production of PE was stopped at a genetic level*. This switching off of PE production accelerated the production of molecular species that cause age-related damage to cells, and thus eventually cell death. When the PE production machinery has not been switched off, cells can recognise when this damage is happening and engage in a process called autophagy, that removes damaged components and replaces them.
This is rather a contrast to what appears to happen in humans, in a condition where there is too much PE. Obesity-induced insulin resistance, the principle form of type II diabetes, correlates with a shift in the lipid profile in erythrocytes [2], including an increase in PE. The amount of PE is reduced upon exercise-induced weight loss. It is not clear from the study whether underweight mammals have too little PE, but there is evidence that autophagy is stronger when PE is higher in mammalian cells [1]. There is clearly a balance to be struck between too much and too little PE.
This balance may not exist for all species, however. PE typically makes up >70% of the phospholipid fraction of the cell envelope of E. coli cells, and so one might think that it is really rather important. However, there is evidence that although growth is not as strong as in normal E. coli, it is possible for cultures to grow with membranes consisting of only the other lipids that would usually be there, phosphatidyl glycerol (PG) and cardiolipin (CL)[3, 4]. The only absolute requirement in this situation is for the presence of divalent cations (doubly-positively-charged ions) such as calcium and magnesium (Ca++ and Mg++) in the growth medium [3]. This is because in the presence of the cations, the CL causes the membrane to curve in the same way the PE would make it do. Thus, the job of the PE as a lipid that elicits membrane curvature, is replaced. However, I think that the fact that the cells grow poorly despite this suggests that this lipid has other functions in E. coli than merely a bulk lipid.
This is certainly the case in mammalian retina cells. The primary amine group in PE is used in the management of de-oxidation of 11-cis-retinal [5]. PE forms an imine with the aldehyde group, that anchors the retinal to the membrane. Various drugs have been tested that mimic this activity, providing some hope for patients with blinding illnesses [6] (and also features in an earlier post).
Aside from this more chemical use of PE, it can also be used as a raw material for a biological action. It can be used as a carrier of ethanolamine, for producing a signalling molecule, N-acyl ethanolamine. The steps in this process are (i) PE is acylated with a fatty acid, (ii) the phosphate di-ester to is hydrolysed to give phosphatidic acid (PA) and the signal, the acylated ethanolamine. When the fatty acid is arachidonic acid, the final signal is called anandamide. The second step is carried out by a phospholipase D (PLD) enzyme [7].
This rather bewildering set of studies invites a number of general conclusions. PE can be, and is, used for some quite different functions, that controlling it is important, and that it has distinct chemical, biological and physical roles. The remaining question is whether this is the end for research into this lipid, or the beginning. Have we found out all there is to find out, of shock value at least, or are there other secrets—is this the tip of the iceberg?
It certainly seems that iceberg lettuce is no way to improve your PE levels, at least for snails. At least two species of snail show that those fed hen’s eggs have higher PE levels by about 40% than those fed only lettuce [8, 9]. Let us hope that the pace of finding out more about this exciting lipid proceeds faster than the average snail.
References
* The gene that codes for the enzyme that produces PE from its precursor, phosphatidylserine, was removed from the organism
[1] P. Rockenfeller, M. Koska, F. Pietrocola, N. Minois, O. Knittelfelder, V. Sica, J. Franz, D. Carmona-Gutierrez, G. Kroemer and F. Madeo, Cell Death Differ, 2015, 22, 499-508. 10.1038/cdd.2014.219.
[2] M. Younsi, D. Quilliot, N. Al-Makdissy, I. Delbachian, P. Drouin, M. Donner and O. Ziegler, Metabolism, 2002, 51, 1261-1268. 10.1053/meta.2002.35184.
[3] J. A. Killian, M. C. Koorengevel, J. A. Bouwstra, G. Gooris, W. Dowhan and B. de Kruijff, Biochimica et Biophysica Acta , 1994, 1189, 225-232. http://dx.doi.org/10.1016/0005-2736(94)90069-8.
[4] A. DeChavigny, P. N. Heacock and W. Dowhan, Journal of Biological Chemistry, 1991, 266, 5323-5332.
[5] M. Zhong, L. L. Molday and R. S. Molday, Journal of Biological Chemistry, 2009, 284, 3640-3649. 10.1074/jbc.M806580200.
[6] A. Maeda, M. Golczak, Y. Chen, K. Okano, H. Kohno, S. Shiose, K. Ishikawa, W. Harte, G. Palczewska, T. Maeda and K. Palczewski, Nat Chem Biol, 2012, 8, 170-178. 10.1038/nchembio.759.
[7] Y. Okamoto, J. Morishita, K. Tsuboi, T. Tonai and N. Ueda, Journal of Biological Chemistry, 2004, 279, 5298-5305. 10.1074/jbc.M306642200.
[8] J. L. Schneck, B. Fried and J. Sherma, Veliger, 2003, 46, 325-328.
[9] K. R. Sousa, B. Fried and J. Sherma, Journal of Liquid Chromatography, 1990, 13, 3963-3972. 10.1080/01483919008049582.
]]>
Mental illnesses are some of the most difficult to define and diagnose. There are at least two scientific and medical/clinical disciplines devoted to it, a number of specialised hospitals, a wealth of mainstream prejudice and a long history of investigation and experimentation—and yet it is an area that is still regarded as one of the most mysterious regarding human sickness.
One treatment that relies upon lipids, and in particular membranes, has been around for some time. Lithium carbonate or citrate can be taken, leading to (positively charged) lithium ions entering the bloodstream. This is useful because they cannot pass over the membranes of neurones, increasing the net positive charge outside the cell. This changes the electronic potential across the membrane, and so changes the threshold at which neurotransmitters such as glutamic acid and serotonin, are released. This has been shown to have a direct impact on the mood of patients with serious mental illnesses.
The question I am curious to answer, in the view of that evidence, is why should those membranes need it, where others do not? Is there something different about the membranes of someone with mild (type II) bipolar disorder, someone with serious and incurable paranoid schizophrenia and someone with an anxiety condition?
Several recent papers in the scientific literature suggest that there may be, and so tantalisingly for sufferers, some of the mystery of mental illness may be in the process of being dispelled. The most recent work was focussed on shifts in the lipid profile of cells in different parts of the human brain with respect to serious mental illness.
Faria et al. [1] have shown that stress-induced depression in mice actually changes the lipid profile of membranes across the whole brain. The levels of phosphatidyl inositol (PI) fall by about 50%. This is made up for by slight increases in bulk lipids phosphatidylethanolamine (PE) and phosphatidylcholine (PC). Although there is evidence that a form of PC has a crucial role in brain function (Nguyen et al. [2], Ben-Zvi et al. [3]), it cannot replace PI in its functions—PI has been found to be significant in cell division but it is also the precursor to several important signalling lipids. However, when the pool of starting material is diminished by half, we might expect that the scope for normal signalling in the brain is compromised.
Although this points towards a biological effect of changing the lipid profile, the fact that it has changed at all raises several questions. What are the physical properties of the membrane after the change in lipid profile? Is the adaptation to the stress stimulus effective? The question I am interested in is the more precise effects. What this study shows is that the result of an emotional experience of a mammal has a direct effect on the molecular profile and thus the function of its brain, but it does not show what the shifts are within the tissues of that organ, or within the organelles of those cells.
This work is therefore an excellent basis for further research. However, it would be disingenuous to consider it only in those terms. This work can be used to inform the understanding we have about the mode of action of certain anti-depressants. Aboukatwa and Undieh [4] showed that the fraction size of a lipid called CDP-diacylglycerol, and PI were both increased on treatment with imipramine, paroxetine and maprotiline (also in mice). This is significant because CDP-diacylglycerol is the precursor to PI, and thus the whole production of inositides is increased by these drugs.
This exciting connection is marvellous grist to the mill of those interested in the behaviour of inositides in biological systems (viz. this author, [5, 6]), but it is also a good connection to another, well-defined set of mental conditions, anxiety disorders. Gormanns et al. [7] used a systems biology approach to explore the changes to PI signalling pathways that were observed in certain psychiatric disorders. They found that dysregulation of the PI signalling pathway was the result of changes in the transcriptome in anxiety disorders.
This fits rather well with the observations about stress-induced changes in the size of the PI fraction described above [1] and thus contributes to an exciting set of studies that link inositides with mental illnesses. It begins to provide a context for older observations about changes in inositides in schizophrenia patients [8, 9]. What is less clear from a clinical point of view is the therapeutic target. Should a particular part of the brain be targeted? How can this be achieved? I predict that analyses of individual parts of the brain will give further insight, along with individual tissue types within those. The data that emerges can then be used to inform our understanding of the underlying molecular mechanism, and thus provide the probable explanation for, psychiatric disorders. Prejudice may yet be beaten by phosphatidylinositol.
References
[1] R. Faria, M. M. Santana, C. A. Aveleira, C. Simones, E. Maciel, T. Melo, D. Santinha, M. M. Oliveira, F. Peixoto, P. Domingues, C. Cavadas, M. R. M. Domingues, Neuroscience, 2014, 273, 1-11. http://dx.doi.org/10.1016/j.neuroscience.2014.04.042
[2] L. N. Nguyen, D. Ma, G. Shui, P. Wong, A. Cazenave-Gassiot, X. Zhang, M. R. Wenk, E. L. K. Goh, D. L. Silver, Nature, 2014, 509, 503-506.
[3] A. Ben-Zvi, B. Lacoste, E. Kur, B. J. Andreone, Y. Mayshar, H. Yan and C. Gu, Nature, 2014, 509, 507-511.
[4] M. A. Aboukhatwa, A. S. Undieh, BMC Neuroscience, 2010, 11, 10. DOI: http://dx.doi.org/10.1186/1471-2202-11-10
[5] S. Furse, N. J. Brooks, A. M. Seddon, R. Woscholski, R. H. Templer, E. W. Tate, P. R. J. Gaffney, O. Ces. Soft Matter, 2012, 8, 3090-3093. DOI: http://dx.doi.org/10.1039/c2sm07358g.
[6] S. Furse, L. H. Mak, E. W. Tate, R. H. Templer, O. Ces, R. Woscholski, P. R. J. Gaffney, Organic and Biomolecular Chemistry, 2015, Advance article. DOI: http://dx.doi.org/10.1039/c4ob02258k
[7] P. Gormanns, N. S. Mueller, C. Ditzen, S. Wolf, F. Holsboer, C. W. Turck, Journal of Psychiatric Research, 2011, 45, 973-979. DOI: http://dx.doi.org/10.1016/j.jpsychires.2010.12.010.
[8] I. Das, M. A. Essali, J. de Belleroche, S. R. Hirsch, Schizophrenia Research, 1994, 12, 265-268.
[9] O. Fedorenko N. Strutz-Seebohm, U. Henrion, O. N. Ureche F. Lang, G. Seebohm, U. E. Lang, Psychopharmacology, 2008, 199, 47–54. DOI: http://dx.doi.org/10.1007/s00213-008-1095-x
]]>
Tweet
Sickle-cell anaemia and brittle bone disease are both caused by a single, small change in the structure of a protein. Cystic fibrosis is also caused by one of several ‘point’ mutations in the structure of just one protein. The question a scientist might ask is why this should occur—why should a small change in structure result in such a striking change in behaviour?
A practical difficulty in answering this question is how to test for the functional difference between normal and disease mutant forms of a protein. Cytosolic proteins can be characterised in aqueous systems that are effectively a model of the liquid part of cells. This means that the protein that is at fault in sickle cell anaemiacs can be characterised reasonably easily. The functional behaviour of the protein in cystic fibrosis is more difficult to characterise because it is a membrane protein rather than a cytosolic one.
Like lipids, membrane proteins have a part that likes water (hydrophillic) and one that does not (hydrophobic). This means that on contact with water without a membrane, they can re-fold, i.e. morph into a bit of a mess. The difficulty with a lot of membrane proteins—both disease and normal types—is that isolating them so that they can be characterised is an insurmountable obstacle.
The practical difficulty is therefore to isolate the protein in its native protein conformation. Detergents have been used for this purpose, and these do go some way to satisfying the amphiphilic character of membrane proteins. However, they can twist them in other conformers, and that can be just a difficult to understand, and just as useless in researching a disease. A better idea is surely to isolate the proteins with a patch of membrane that they like, such as the one they have naturally?
This is exactly what several groups of scientists have done [1-3]. The heart of this technique is to use a sort of molecular belt that acts like a polymerised lipid, with the greasy part of the lipid facing the fatty acid residues of the lipids and the hydrophilic part of the polymer facing the aqueous fraction. It turns out that this is thermodynamically stable and that sections of this polymerised lipid of the right length can behave like pastry cutters and chop out little discs of the membrane, with the all-important membrane protein nonchalantly sitting in the middle, like a prince in a sedan chair (Figure).
Figure. A section of the membrane is separated from the rest by the addition of co-polymer (yellow) to give a nanodisc (bottom).
Long et al. [1] showed that several mitochondrial proteins could be isolated this way. They established this by showing that the proteins isolated still had their enzyme activity after the isolation. This provided excellent data that functioning proteins resident in the membrane can be isolated for studies conducted in vitro.
Swainsbury et al. [2] tested different membrane protein isolation methods using a photoreaction centre from a purple bacterium called Rhodobacter sphaeroides. This protein complex is a useful tool for comparing methods because its behaviour with respect to light is dependent upon its conformation. In fact, both small and large differences in the conformation of the protein can be measured.
Doerr et al. [3] chose a different and more difficult target, an ion channel. Ion channels transport metal ions like sodium and potassium across membranes. The difficulty in isolating and characterising these is that unless they are in a membrane that divides two aqueous compartments, the changes in concentration of the ions cannot be measured. It is the molecular equivalent of letting someone in by opening the door of a room that has a wall missing. What this group therefore did was to isolate the ion channel protein complex (called KcsA) and reconstitute it in a membrane that did indeed divide two aqueous compartments. Electrophysiology techniques were then used to establish the activity of the KcsA they isolated. They found that indeed it was active after being both put into these little membrane patches (nanodiscs) and then reconstituted into another membrane.
What this research shows is that a variety of proteins, with different functions and from different species, can be isolated and characterised using this approach. The work is at an early stage, meaning that there is the unfortunate but predictable rush to secure the Intellectual Property rights. One form of this is a registered trade mark that has suffered from 90s-style marketing, having the name ‘lipodisqs®’. This has also meant that the material for making nanodiscs has become commercially available. It is at a high price, the equivalent of £6m/Kg in the Netherlands.
However, as the facility of this technique becomes clear and more widely used, I predict that it will become easier to use and more finely tuned. We may yet see the characterisation of membrane proteins that exist in tiny quantities, such as those in individual neurone synapses. It may allow us to determine shifts in membrane protein concentration at different points in the cell division cycle. This technique may have scope to allow us to chart the shifts in molecular profile that represent the processes of a normal working body. However, its strongest application in the current zeitgeist may be to determine the functional difference between individual membrane proteins in diseased and healthy systems, enabling the characterisation of disease in a clearer and quicker way than ever before.
References
[1] A. R. Long, C. C. O’Brien, K. Malhotra, C. T. Schwall, A. D. Albert, A. Watts, N. N. Alder, BMC Biotechnology, 2013, 13, 41. DOI: 10.1186/1472-6750-13-41
[2] D. J. K. Swainsbury, S. Scheidelaar, R. van Grondelle, J. A. Killian, M. R. Jones, Angew. Chem. Int. Ed., 2014, 53, 11803. DOI: 10.1002/anie.201406412
[3] J. M. Doerr, M. C. Koorengevel, M. Schäfer, A. V. Prokofyev, S. Scheidelaar, E. A. W. van der Cruijsen, T. R. Dafforn, Marc Baldus, and J. A. Killian, Proc. Natl. Acad. Sci., 2014, DOI 10.1073/pnas.1416205112.
Conflict of Interest Statement: I share an office with the first author of [3]. The last author of the same paper is my boss, though I work on a separate project.
]]>
A sturdy poetic metaphor for morning frost on grass is it being as dusted in sugar. This is a prescient analogy, given the data that is emerging about how organisms that survive the low temperatures, and desiccation.
How some living organisms can survive harsh cold or drought for extended periods, where others expire with only a modest drop in temperature or water availability, has been a puzzling question in biology for some time. There are several surprising examples–compare the story of some seeds that were found in the pages of a diary that had not been touched in the 17th century that were germinated and grown successfully at Kew Gardens, with a variety of fruiting plants that are susceptible to frost and need special attention when colder weather approaches. Summer fruit like plums are amongst the most susceptible.
There are also rather ordinary examples. A surprising number of the seeds with which we are familiar, are desiccated–sunflower, rape, mustard, hemp, sesame, poppy, hazelnut, walnut, acorn, and so on. The inescapable question is how they survive being put into what amounts to a static existence. Further questions come to mind almost immediately–how do cells in stasis know when to re-animate, how do they go about doing so once they have started? In order for this process to work, a number of individual cells must work in a co-ordinated way so that the organism can bounce back into active life in a controlled way.
The first question, of how being put into dry or frozen stasis without causing death, it is a good question from a molecular perspective as well as an ecological one. Lipids, and thus cell membrane studies, are central to this. We know from lipid physics studies that when the temperature falls below a certain point, the packing of the alkyl fraction of lipid bilayers changes in a way that heavily compromises its barrier properties. Significant drops in temperature can crystallise membranes.
A recent paper by Konov et al. [1] gives us some clues as to how scientists are beginning to understand resistance to these changes in lipid packing. In some recent work [1], they investigate the mediating effect of sugars (e.g. sucrose, trehalose) and sugar alcohols (e.g. sorbitol) on the behaviour of model lipid bilayers. They showed that these sorts of molecules stabilise the bilayers we see under ambient conditions, so that they remain in place at lower temperatures.
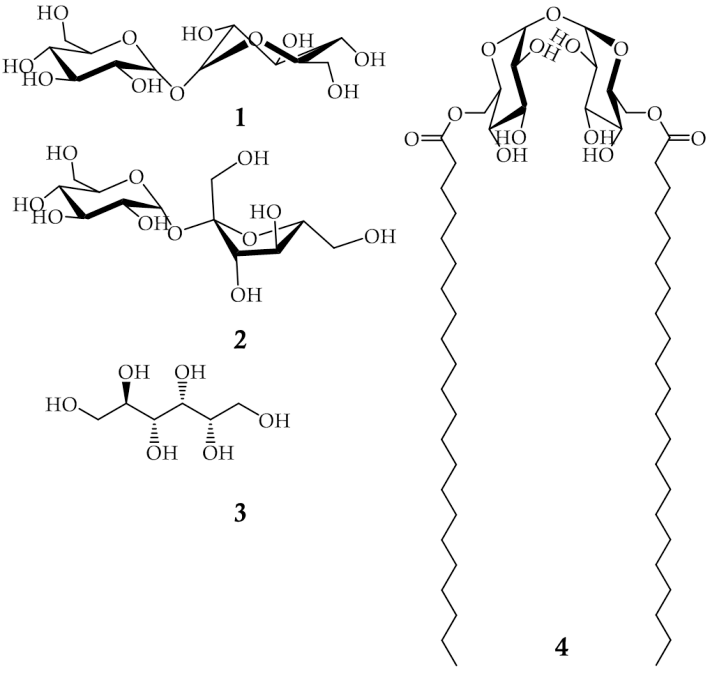
Figure. Trehalose (1, top left), surose (2, middle left) and sorbitol (3, bottom left), trehalose dibehenate (4, right).
What this group of authors conclude is that these sugars have the ability to replace the water molecules that interact with the bilayer under more ambient conditions. The molecular structure of the water and sugars are a clue to this (Figure). This means that the membrane behaves much more like it would do when water is present. This in turn has led to such molecules being called ‘cryoprotective’ reagents.
There are several biophysical techniques that could have be used to test the hypothesis that sugars are cryoprotective agents. In principle, any biophysical technique that can be used to indicate transitions between phases as a function of temperature could be suitable. A phase transition indicates a change in the way the lipids are assembled and thus moves away from the ‘normal’ membrane. In this work, they used a technique called Electron Spin Echo (ESE) Spectroscopy.
ESE is a relatively new technique. It is described as a pulsed version of Electron Paramagnetic Resonance, which means that it detects orientational vibrations (librations) on the nanosecond time scale [1,2]. It does this only in a particular type of molecular species, one with an unpaired electron, called a free radical. Specialised molecular probes, called nitroxide labelled stearic acid, are used in these systems at low concentrations. In this work [1], it has given clear evidence that certain molecules (sucrose, trehalose) are beer cryoprotective agents than others (sorbitol).
What is not yet clear is what happens on thawing, or in more complicated systems. In order to understand the full process of freezing, or desiccation, on an organism, what happens during re-animation is also required. We also have yet to know what happens to membrane systems that comprise membrane proteins, like ion channels or hormone receptors. The tools for research in the direction of controlled desiccation/freezing, and re-animation, may be closer at hand. A membrane-form of trehalose is commercially available, called trehalose dibehenate (Figure, R). The fatty acid residues on this sugar mean that it will be thermodynamically favourable for this species to be located in the membrane. This guarantees that a number of trehalose molecules will be at the surface of the membrane, irrespective of the prevailing conditions. The presence of such a species on the membrane may confer stability during desiccation and freezing.
The exciting thing about work in this field is that we know the process happens frequently in nature, and that understanding it is the first step to harnessing it, probably with incalculable results.
References
[1] K. B. Konov, N. P. Isaev, S. A. Dzuba, J. Phys. Chem. B, 2014, 118, 12478. DOI: 10.1021/jp508312n
[2] N. V. Ivanisenko, S. A. Dzuba, Appl. Magn. Reson., 2013, 44, 883−891. DOI 10.1007/s00723-012-0436-4.
]]>
An analogy of lipids I often use is that they are cellular bricks. They make up the membrane, the wall that separates the inside of the cell from the outside. So, this explanation gets across many of the facets of the lipid quite concisely—there are a variety of sorts, they do not work as individuals, when assembled together they form stable and often lasting structures that are usually much thinner than the spaces they enclose. The epithet ‘cellular bricks’ also suggests that, at least for individuals, they are not that exciting.
I make light of this by tackling the ennui head on, and use the word boring. This gets a half-laugh and gives my interlocutor the chance to say whatever is on their mind, usually either desperately thinking of something to say about anything else, or processing the idea that the word lipid is not synonymous with the word fat.
The trouble with this explanation is that it often leaves me feeling as though I have been pessimistic. There is no proper ‘lift’ at the end to leave things on a nice note.
I am pleased to say that recent work by Kuge et al. [1] and Ersoy et al. [2], has made me change my ideas about how I describe basic lipids—or at least reconsider my stand-up comedy explanation of them. These two reports show that one of the most common lipids in mammals and yeast, phosphatidylcholine, may be rather more exciting as a biological molecule than previously expected.
Kuge et al. [1] show that a particular type of PC, called 1-oleoyl-2-palmitoyl phosphatidylcholine, is concentrated at the tips of growing nerve cells. Ersoy et al. [2] report evidence that an enzyme called PC-transfer protein has a role in insulin signalling. Specifically, PC-transfer protein is involved in a part of the process that goes wrong in type II (obesity and gestational) diabetes.
These two things show a breadth in the functions of PC that are a considerable step forward in our understanding of this lipid.
Delving into it further, I found that there have been hints about this other life of PC for a while. The first evidence appears to be that PC can be a storage lipid. Specifically, it is a pantry for a kind of poly-unsaturated fatty acid called [3,4] that is itself a starting material for prostaglandins in inflammation pathways. Another nudge towards Bethlehem was the observation that it is used to make sphingolipids, lipids that are themselves important signals [5] (you can read more about the importance of sphingolipids here [6]).
The role of PC as a protagonist in the mechanisms behind cancer make it a worthy research interest. However, could one argue that the physical role of PC is more fundamental to our cells?
The first focussed biophysical work on lipids was done on PC in the 1960s. This early work was dominated by Vittorio Luzzatti, who published a paper in 1968 called ‘Polymorphism of Lipids’ [7], and much of it was done on PCs. It was shown that PC has a strong thermodynamic preference for forming bilayer sheets. This correlates almost exactly with how our cellular membranes are constructed. Cellular systems differ in that they have a gentle curvature, that is what allows them to be spheroid objects. In terms of bilayer geometry, this is easily accounted for, just have fewer PC molecules on the inside leaf. Added to which, there are other lipids present anyway. Several of the latter turned out to be lipids that induce curvature in membranes.
These two bodies of work, what we know about the physical behaviour of PC in forming membranes and its biological role in cancer and diabetes, are difficult to compare. It is almost the lipidology equivalent of fatuous questions about whether Newton or Shakespeare is better, or whether a sound exists if a person has not heard it.
The way I prefer to think about it is that the physical and biological importance of PC is like comparing the colour of an orange with its flavour. They are separate things, distinct aesthetic experiences. And one without the other is a bit spooky.
References
[1] H. Kuge, K. Akahori, K. I. Yagyu. Journal of Biological Chemistry, 2014, 289, 26783. doi: 10.1074/jbc.M114.571075
[2] B. A. Ersoy, A. Tarun, K. D’Aquino, N. J. Hancer, C. Ukomadu, M. F. White, T. Michel, B. D. Manning, D. E. Cohen. Science Signaling, 2013, 6, ra64. doi: 10.1126/scisignal.2004111
[3] V. A. Ziboh, J. T. Lord, Biochemical Journal, 1979, 184, 283.
[4] R. M. Kramer, D. Deykin, Journal of Biological Chemistry, 1983, 258, 13806.
[5] W. D. Marggraf, F. A. Anderer, Hoppe-Seylers Zeitschrift Fur Physiologische Chemie, 1974, 355, 803. doi: 10.1515/bchm2.1974.355.2.803
[6] B. Ogretmen, Y. A. Hannun, Nature Reviews Cancer, 2004, 4, 604. doi:10.1038/nrc1411
[7] V. Luzzati, A. Tardieu, T. Gulik-Krzywicki, Nature, 1968, 217, 1028. doi:10.1038/2171028a0.
]]>
The best word I can find for my reaction to a shoulderful of dandruff is off-putting. I have yet to meet anyone whom I know would disagree, including those who sport a pair of such shoulders themselves on a daily basis (if only they knew it). The only further thought I had had until now was an unkind one about whether or not those afflicted with this condition, use an anti-dandruff shampoo.
It is common knowledge that the white particles that we recognise as dandruff are flakes of scalp, i.e. the skin out of which our head-hair grows. The proper name for the condition is seborrheic dermatitis and it is something that affects 20% of humans of all ages. It is similar to what is called atopic dermatitis, a condition in which the non-scalp skin flakes off. It is often known as eczema. Atopic dermatitis affects 10-20% of children, but typically many fewer adults.
What I had never considered was what might cause the dandruff in the first place.
Recent research by Ishikawa et al. [1], Janssens et al. [2], Park et al. [3] and Tawada et al. [4], provide some clues. Taken together, these reports shows that the occurrence of atopic and seborrheic dermatitides correlate with a shift in the lipid profile of the skin. The evidence suggests that one lipid (with fatty acid residues that are 16 carbon atoms long, C16) is gained at the expense of one that is longer (24 carbon atoms long, C24). In other words, the hypothesis these data led to was that the fatty acids used to make these lipids are not made long enough.
In order to make sense of these observations scientifically, a recently-publsihed article by Skolova et al. [5] described the use of used a model system to research the physical impact of this change. This model was used whilst ensuring that the data generated were relevant to human skin (stratum corneum).
The model system they used consisted of the ceramide (either C16 or C24), a fatty acid (of typical chain length for mammalian lipid systems, C16) and cholesterol, also normally found in the stratum corneum. They determined the physical behaviour of the system in a comparable way using infra-red spectroscopy. This type of spectroscopy demonstrates how the fatty part of the lipid systems pack together. This technique can therefore be used to investigate the differences in packing between the C16 and C24 systems.
Both model systems behave a bit like ordinary bilayers, with the hydrophobic effect ensuring that bilayer-type systems are formed. However, the additional length of the C24 fatty acid residue in the ceramide may lead to several significant differences in the physical properties and parameters of the system. For example, the bilayer may thicken. Alternatively, the longer chain of the fatty acid residue might bend or even fold, to fill in gaps in curved systems.
In this system, which is typically rather drier than most naturally-occurring lipid systems, the effect is somewhat different. The evidence suggests that instead of a classical bilayer, the ceramide behaves more like a wax, in which the carbon chains are 180 degrees apart, rather than parallel (blue molecule, Figure). This means that the head group of the ceramide represents the hydrophilic part of the system—that which would normally be two faces of head groups and an aqueous fraction—and the system is held together as much by the van der Waal’s forces between hydrophobic moieties as it is by the hydrogen bonding and electrostatic (charge-based) forces.
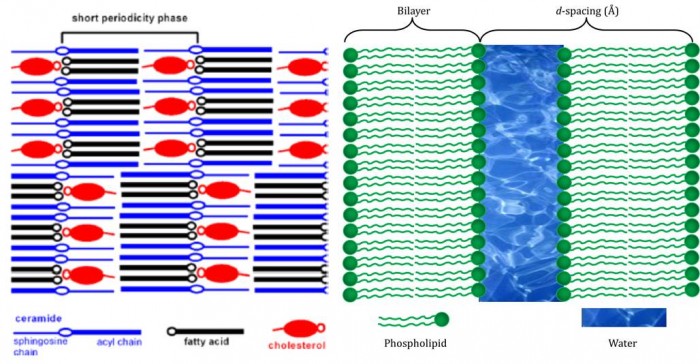
Figure. Left: diagram of the molecular assembly of ceramides, fatty acids and cholesterol in human skin. The blue molecules are not packed as classical lipids, but splayed at 180°, leaving the head group (blue ‘O’)in the middle, rather than on one face. Note how the hydrophilic parts of the cholesterol (red ‘o’) and fatty acid (black ‘o’) are assembled facing one another adjacent to that of the ceramide, forming an hydrophobic surface. (this diagram is from Skolova et al.[5], from data published by Iwai et al. [6]. Right: An ordinary bilayer. The structural parameter marked as ‘d-spacing is used to record the size and shape of systems.
It is therefore straightforward to realise how a much shorter chain—sorter by a third—might interfere with the balance of forces present in the system. We therefore expect that the skin that flakes to produce dandruff is rather weaker than normal and is the result of a faulty enzyme. The enzymes that produce longer-than-average-length fatty acids are called elongases.
Despite this insight, and how it has changed my view of what dandruff is, I must confess I cannot find use for it in conversation. Would anyone be able to say to someone with eczema “I’m sorry to see your elongases are not working at the moment”?
References
[1] J. Ishikawa, H. Narita, N. Kondo, M. Hotta, Y. Takagi, Y. Masukawa, T. Kitahara, Y. Takema, S. Koyano, S. Yamazaki, A. Hatamochi. Journal of Investigative Dermatology, 2010, 130, 2511−2514.
[2] M. Janssens, J. van Smeden, G. S. Gooris, W. Bras, G. Portale, P. J. Caspers, R. J. Vreeken, T. Hankemeier, S. Kezic, R. Wolterbeek, A. P. Lavrijsen , J. A. Bouwstra. Journal of Lipid Research, 2012, 53, 2755−2766.
[3] Y. H. Park, W. H. Jang, J. A. Seo, M. Park, T. R. Lee, Y. H.Park, D. K. Kim, K. M. Lim. Journal of Investigative Dermatology, 2012, 132, 476−479.
[4] C. Tawada, H. Kanoh, M. Nakamura, Y. Mizutani, T. Fujisawa, Y. Banno, M. Seishima/ Journal of Investigative Dermatology, 2014, 134, 712−718.
[5] B. Školová [Skolova], K. Hudská, P. Pullmannová, A. Kováčik, K. Palát, J. Roh, J. Fleddermann, I. Estrela-Lopis, K. Vávrová. Journal of Physical Chemistry B, 2014, 118, 10460 doi:10.1021/jp506407r
[6] I. Iwai, H. M. Han, L. den Hollander, S. Svensson, L. G. Öfverstedt, J. Anwar, J. Brewer, M. Bloksgaard, A. Laloeuf, D. Nosek, S. Masich, L. A. Bagatolli, U. Skoglund, Lars Norlén. Journal of Investigative Dermatology, 2012, 132, 2215–2225;. doi:10.1038/jid.2012.43.
]]>
Biological systems have a constant need for merging lipid bilayers.
This statement sounds counterintuitive at first glance: cell replication and endocytosis, the process by which white blood cells kill threatening bacteria, is the division of membranes. Why should there be value in fusing them?
The answer is surprising and ordinary. The principal example is a process that happens far more times per day than the others. The best example of our need to fuse membranes is the transmission of nerve impulses across nerve synapses. Nerve synapses are the junction boxes between two nerve cells that pass an impulse from one to the next. A good example is a simple reflex, like the connection between the thermoreceptors in our fingers and the muscles in our arms that jolt them away from the heat.
This seems a good enough reason on its own, for our bodies to have this process well worked out. However, physical chemistry is not a need-based subject: bilayers are energetically favourable, that is what drives their formation in the first place. So, what can make this process happen at the speed of a nerve impulse?
One answer is a kind of bridging protein, known as SNARE. This is one of the less tragic and more tasteful acronyms in science (compare with the organic chemists who are trying to push for diethyl azodicarboxylic acid to be referred to as DEAD). SNARE stands for Soluble NSF Attachment Protein REceptor. It is made up of several pieces (See Figure).
Figure. The fusion of a vesicle to a cellular membrane (labelled ‘target’ in top picture). v-SNARE, the part of the system that is attached to the vesicle at step one, interacts with t-SNARE, the part of the system on the target bilayer. The connection between these two draws the two bilayers near one another, before docking occurs. This is followed by hemi-fusion, in which the exposed monolayers of the vesicle and target begin to merge, and is followed by fusion, in which the aqueous fractions of the compartments begin to mix. Image courtesy of the University of Tennessee, http://www.bio.utk.edu/BCMB513-1/bcmb513-1.htm.
According to Thomson Reuters Web of Knowledge, the first paper in which the authors dared, and were allowed, to use this appealing acronym was published in 1994. Those authors, Sogaard et al. [1], describe a protein, called Rab, which is required for the assembly of SNARE complexes.
Other proteins, called chaperones, are required for the proper activity of SNARE [2]. This process is regulated by some of the lipids in the bilayers that are being fused: sterols, diglycerides and inositides. This implies that the membranes being fused are ‘consenting’ to being so, as without the correct mix of these lipids, the process cannot take place.
A measure of agreement by the owners of both bilayers seems fair, as the process is very quick on the scale of biological activity. Liu et al. report that it can take just 25 milliseconds [3]. That is rather less than the blink of an eye – a process that requires at least one such fusion, to occur.
What is less well understood, but has been observed in some detail is a sort of leakage that occurs during the process. It seems that the membrane properties of the two bilayers are shifted considerably with respect to their normal behaviour, as the leakage of small molecules from one or both bilayer-bound compartments during the process, can be measured [4].
We therefore know much about the process of fusing bilayers. The protein machinery, the speed at which it occurs and concurrent processes are documented. This might seem as though all of the questions have been answered and that we are but a hair’s breadth from closing the book on SNAREs. Not so: at the time of writing, the most recent work* on SNAREs to be published concerns the computational modelling of SNARE proteins, in which the molecular dynamics of a neuronal SNARE protein have been explored [5].
As well as understanding millisecond processes that we take for granted – they are of course the ones relied upon for me to type this and for you to read it– the physics of individual macromolecules can be explored as well. Perhaps only time will tell how much has yet to be enSNAREd.
References
[1] M. Segaard, K. Tani, R. R. Ye, S. Geromanos, P. Tempst, T. Kirchhausen, J. E. Rothman, T. Soellner, Cell, 1994, 78, 937.
[2] J. Mima, C. M. Hickey, H. Xu, Y.Jun, W. Wickner, EMBO J., 2008, 27, 2031.
[3] T. Liu, W. C. Tucker, A. Bhalla, E. R. Chapman, J. Weisshaar, Biophys J., 2005, 89, 2458.
[4] P. C. Zucchi and M. Zick, Mol. Biol. Cell, 2011, 22, 4635
[5] M. Tekpinar, W. Zheng, Journal of Molecular Modelling, 2014,
20, 2381.
*Retrieved 27th August, 2014.
I babysat for a paedophile when I was a teenager. Twice. I did not know he was at the time, though it began to emerge in the months afterwards. First, he was accused of assaulting a boy who was in his early teens. It was hushed up. He and his wife were friends of my parents, and wanting to be supportive, my mother had invited them over. I remember he looked very uncomfortable. I know now that what he was feeling was probably guilt and embarrassment rather than fear.
A few months later he was accused again. The alleged assault was a more serious one this time, and with having been accused before, it had to be looked in to. I suppose it must have been pretty clear the second time as it was not hushed up.
I realise and recount this because of the stream of sexual-abuse-of-children stories in the news recently, in Britain at least. Cases cover a range of prominent people, celebrities, public figures and even former members of parliament have been implicated. Several of those mentioned are dead, limiting the scope for any kind of retribution. There have also been cases of children’s homes at which child abuse seems to have been almost cultural, in Rotherham and Belfast.
There was no hiding in plain sight with the paedophile I knew. No fake gold medallions. No creepiness, nothing obnoxious or unpleasant. He had a moustache I thought was strange, but I’ve never seen one I liked so I suppose that is just my preference. He was a musician and music teacher.
He must have known it was not a good idea to seek sexual gratification with a child. I suppose he wanted for a moment’s carnal pleasure, but rationally he must know that indulging it would destroy the life he had carved out for himself. He must have known that sex that was not consented to by both partners–which is what shagging choirboys boils down to if the emotional effect is stripped away–would land him in at least a dozen types of trouble. Touching children is about the most reprehensible crime around these days, followed closely by sex without consent with an adult, so the chances of re-carving out the same life again afterwards were virtually nought.
It does not take much emotional understanding to realise that he must have had these feelings for a long time. Presumably it develops like hetero- or homosexuality and reaches fruition in teenage years. Observation from what gets reported suggests that paedophiles seem to have a preference for boys in their early teens or girls in the few years after they go to school. In other words, embryonic masculinity or embryonic femininity, framed with a certain innocence.
Paedophilia has been around for a long time, and so it is unrealistic to expect it to vanish any time soon. Someone who fancies children, and perhaps always has, probably always will. Just like fancying men or women. The question on my mind is what we do about it.
The rape part is relatively straightforward to manage. We have a legal process for that, for example the Sexual Offences Act (2003). I think it is also fair to say that there is not the political will for it to be modified substantially in the next few decades. I do not advocate changing it. Forced sex with someone who does not understand what it is, or expect it, and is not yet able to make the decision about whether it is what they want, is wrong and can be damaging.
A less easy question is about what happens outside of the rape. What about someone who has those feelings but who has never ‘touched’ a child in his or her life? Is it safe for them to be around children? I fancy women and it is considered safe for me to walk past a beach on a warm summer’s day. Is it safe for an equivalent man in his 30s who fancies children to go to work at a school as a builder, say, or carpet layer?
Many would say it is not safe. I think this objection is a strong and well founded one. However the argument that it is not unsafe for such an individual to be near children is hard to answer. There is no criminal activity involved in feeling something. Feeling something does not mean any kind of action will follow. These observations make it hard to assess the value of the argument that such individuals should be kept at arm’s length from children. Just saying ‘as long as nothing weird, creepy or illegal happens’ is hardly a workable approach; political correctness means we are not in a position to set guidelines on how people who fancy children should behave.
This means a rational position on this is hard to find, especially when paedophiles seem to be increasing in prominence. Perhaps it is far more common than we like to believe. Homosexuality is commonly understood to be about 10% in human populations and estimates of bisexuality put it at about 30%, with the rest as heterosexual. Where does paedophilia fit in to that?
What I do think is more certain, is that we have seen an about-turn in the last hundred years or so, when it comes to sexuality. The Oscar Wilde trial showed that homosexuality was still a terrible no-no for the Victorians. So infamous was it, that it influenced behaviour between men for years afterwards and still does now. Paedophilia, as far as the evidence we have of that time suggests, was much less controversial in that period. It certainly was not punished in the way it is now.
Ultimately, we cannot change a person’s sexual feelings, nor can we allow them to be expressed when doing so can cause long-term and unnecessary emotional damage. However, unexpressed sexual emotion creates a frustration for those who feel it that may be just as damaging.
A heterosexual or homosexual person can masturbate, perhaps with the use of pornography. They could buy the services of a prostitute, to quench their sexual tension. In terms of what is permitted legally, a paedophile can masturbate with whilst recalling his/her fantasies, but that is about it. That might satisfy a short-term craving, but that is not enough to stop every paedophile from turning their life inside out and forever, because of those feelings. Perhaps the important thing for us to do is understand why a person would be driven to risk so much on a futile sexual encounter.
I never did know what happened to the man I babysat for. He disappeared from the radar, presumably intentionally, in order to start afresh somewhere else. A putative google search suggests that he might have moved to another part of the country, but it is not clear. I wonder if he feels as though he is on the run.
]]>
It’s time for a change. For too long ignorance and made-up superstition have occupied the foreground. Ignorance has been given a place at the top table because of the notion that it represents the ordinary person’s point of view. Superstition is afforded the same if not a greater privilege because the act of creating thoughts based on feelings and imagined beings, is worthy of reverence.
Scientists have tried to demonstrate the reality using reason and evidence. The resulting work has told us a great deal, but the truth is that it is not effective in the wider sense. Not because the evidence is not there, but because science is not a political cause. Politics is not scientific, and so it does seem perverse to adopt it for scientific ends. However, politics is used for a variety of ends that started off having nothing to do with it–feminism, environmental protection, building large numbers of useless tractors.
Politics is a blunt instrument, and so you might ask why scientists should adopt it in order to do science, when they are used to finely-tuned instruments. The reason is that scientific instruments measure, politics gets things done. We see democratic politics achieve all sorts of things that were not predictable and are not sensible. Various groups of people around the world are given open-ended privileges by governments because long-dead members of their minority suffered even longer ago. It is fair to say we have seen a bit of that for scientists, especially women, but that is about it. Who is sticking up for science and scientists as a whole?
There are a few people who get in under the radar — Dara O’Briain for example. He studied Mathematics and Physics as an undergraduate; it was not until he was a hugely successful stand-up comedian that he could get the BBC to make a programme about science. And that only ran for one series.
There are one or two soft or cameragenic scientists who get in under the wire and give science a good name. Professors Jim Al-Khalili and Brian Cox spring to mind. They are nice chaps, but I do not see either of them wielding a political scimitar any time soon. Professor Richard Dawkins is often cited as a defender of science against the bonkers and the ill- or uninformed. He is also accused as someone who likes arguing. (It’s hard to disagree with that when one considers just how verbose he is. I spent three hours reading The God Delusion; I have no doubt all of what I read could be edited down to 20 minutes, even by an untrained editor like me.)
So we have a vacuum precisely of a politico-scientific shape. We have scientists who are trying to chip away at blind and woeful ignorance, but with no political voice. At the last count we had just one research scientist in the House of Commons, but he is a Liberal Democrat, which is rather a shame.
That is not to say under-the-wire approach has been entirely ineffective. The group of scientists who adopted a political strategy, political language and a deferential manner, got the funding and political backing for sequencing the human genome. That was an immense scientific and political achievement. In the short term it has been something of a white elephant, it has yet to lead to scientific discoveries of the same magnitude, or to the political backing for other scientific projects of the same magnitude.
So, it can be done, but the stealth approach will not work forever. It is time for scientists to go from fundamental to fundamentalist.
]]>
Nothing makes a sensational news story quite like one centred on a medical drama. Sex sells insalubrious newspapers, as well as everything from ice cream to motor cars, but it does not have the same impact as a story that pricks the mortality of the reader, or someone dear to them.
Stories about recent question marks over published evidence relating to statins – drugs that lower cholesterol concentrations and can therefore prevent certain types of heart disease – have done just this. A variety of news organisations have reported the story, including more reputable ones aiming at a general audience (BBC reports here and here), but also aimed at the scientific community, ensuring exposure to both trained and probably untrained audiences.
This broad presentation of the story led to my receiving worried e-mails asking whether I think the respondent should continue to take these medications. Needless to say, I cannot possibly advise, but it did set me thinking about what the evidence was behind this story and why there was a debate at all. Why was the evidence about statins not clear enough for senior physicians to make a decision on whom should take them or not?
The root of the problem was that two scientific papers about the side effects of statins. In these papers, whether or not it is worth patients who have a low risk of heart disease taking statins was questioned. In other words, if someone in the lowest risk group with respect to contracting heart disease takes statins, side effects like throat, joint and muscular pain, nosebleeds, and digestive upsets, may be palpable but the benefit(s) of the drugs are not. Because of the newsworthy status of this topic, the papers were discussed in British Medical Journal articles, such as one by Aseem Malhotra.
From a scientific standpoint, the striking thing about these articles and the subsequent commentaries is two-fold. First, the data was not analysed correctly and neither was this oversight noticed by its authors, reviewers or editors. Inevitably, this reflects poorly on all parties involved and taken together, these seem a devastating failure for a high-profile journal, especially one that is also read by readers who are not medically or scientifically trained (if only journalists).
Second, the evidence that was mis-analysed and possibly misinterpreted was data from an uncontrolled observational study. The phrase ‘uncontrolled observational study’ is lumpy and I suspect rather opaque to non-scientists. Clarity about its meaning has been missing from the general debate, so I provide an insight here:
Observational means that the data has been collected by simple measurement from a given cohort, with calculations made thereafter. This cohort is likely to be an homogenous group, and one hopes a large one, so that the differences between individuals that occur ordinarily do not skew the overall result. For example, if you wanted to test a sun cream, you might opt for a group of people that represents the spectrum of those who are very susceptible to sunburn through to those who are much less so, with everything in between. But, you would also want to know that all the individuals spent about the same length of time in the sun, so the recorded values were comparable and made sense when an average was calculated. This is where ‘observational’ is a devastating compromise, as the latter point is not always met, and so the data between individuals varies considerably and in different ways. The point that someone is more susceptible to sunburn than someone else, is therefore lost. Even enormous groups are insufficient for providing a clear answer. The more serious problem with observational studies however is that no test has been carried out. A series of measurements have been taken, but no scientific conclusion can be drawn, because no causal link can be made. Only a correlation exists, because a direct link has not been established.
Uncontrolled is also a serious compromise with respect to the quality of the data and what it means. A controlled study is one in which there are adequate comparison measurements made, in order to show the significance of the data from the test subjects. Using the sun cream example, controls in that study would be people who did not use sun cream at all (or one without the active ingredient) but also ones in which sunburn is known to occur. The latter are called positive controls and are a way of determining what the thing one is looking for actually looks like. There are, or course, ethical concerns about such things, but scientifically, in order to determine whether a given occurrence is the causal result of something, clear comparisons are required in order to assess the significance of the data from the test subjects.
Observational studies, controlled or not, do not have this facility. It is a real shame as many things are not easy to test causally, and the ethical considerations for many others are insurmountable. However, observational studies are just that: they see, they do not determine scientifically, and they should never be given greater credence than that. A great emphasis is placed on ethically soundness in life sciences studies, but we lack the same attention to scientific principles.
]]>
The simplest method for dieting is to eat less and take more exercise. Specifically, to eat less fat and exercise for longer-than-normal periods so that stored fat (triglycerides) reserves are depleted or used up. The reasoning behind this is simple: more energy is used than is stored.
A question that is rarely tackled is why weight loss should be necessary in more than a handful of serious medical cases. Why do we want or need to lose weight in the first place?
Social and cultural factors form the basis for one set of reasons. Eating three times or more a day, eating food prepared by a global, profitable and well-structured industry, means whatever we might want is available at any time of the day or night. It is therefore easy to eat too much.
But what about at a genetic level? Why do some people seem to have the ability to turn food into reserves of energy seemingly easily, and others make muscle? Why is my hunger for beef burgers being fried not in step with the number of miles I have cycled in the previous 24 hours?
A simplistic answer is evolution. Recent research has provided evidence that the inter-breeding between human species several tens of thousands of years ago, led to a distinctive lipid catabolism in modern European populations [1]. The hypothesis is that in Europe, there was a selection pressure that resulted in human populations who stored fat easily surviving, and those who did not, perishing. Further, this selection pressure did not occur in Africa or in the Orient.
This hypothesis was informed by evidence published in the last decade, that shows that between 1% and 4% of the genome of European humans originates from Neanderthal individuals [2-4]. Importantly, a portion of this fraction is dedicated to lipid catabolism, and is one that contemporary African and Oriental humans do not possess. None of these genes are present in chimpanzees.
It has been suggested that the presence of Neanderthal genes is why lipid catabolism is so different between current human populations. It is also why a significant number of Oriental individuals appear to lay down muscle rather than fat, when they over-eat.
Kharmeeva et al. [1] describe the conclusion that the lipid catabolism of contemporary Europeans was the result of positively-selected gene flow from Neanderthals as appealing, stopping short of giving it cast-iron approval. They state that their observations are also consistent with other hypotheses, based on the number of variants of these genes that left Africa when human populations began a global spread.
Whatever the precise reason, the evidence that shows a genetic distinction in lipid catabolism between current human populations, is not in doubt. What might be of interest to an amateur evolutionary biologist, is what the future might hold for human populations, especially if the results of climate change are as catastrophic as some commentators suggest. Will the ability to grow muscle easily be selected for or against? Similarly, will the ability to lay down fat be an advantage or not?
For the moment, I want to find my bike and burn off a few calories.
References
[1] E. E. Khrameeva, K. Bozek, L. He, Z. Yan, X. Jiang, Y. Wei, K. Tang, M. S. Gelfand, K. Prufer, J. Kelso, S. Paabo, P. Giavalisco, M. Lachmann, P. Khaitovich, Nature Comm., 2014, 5, 3584. DOI: 10.1038/ncomms4584
[2] R. E. Green, J. Krause, A. W. Briggs, T. Maricic, U. Stenzel, M. Kircher, N. Patterson, H. Li, W. Zhai, M. H. Y. Fritz, N. F. Hansen, E. Y. Durand, A. S. Malaspinas, J. D. Jensen, T. Marques-Bonet, C. Alkan, K. Prüfer, M. Meyer, H. A. Burbano, J. M. Good, R. Schultz, A. Aximu-Petri, A. Butthof, B. Höber, B. Höffner, M. Siegemund, A. Weihmann, C. Nusbaum, E. S. Lander, C. Russ, N. Novod, J. Affourtit, M. Egholm, C. Verna, P. Rudan, D. Brajkovic, Ž. Kucan, I. Gušic, V. B. Doronichev, L. V. Golovanova, C. Lalueza-Fox, M. de la Rasilla, J. Fortea, A. Rosas, R. W. Schmitz, P. L. F. Johnson, E. E. Eichler, D. Falush, E. Birney, J. C. Mullikin, M. Slatkin, R. Nielsen, J. Kelso, M. Lachmann, D. Reich, S. Pääbo, Science, 2010, 328, 710–722 .
[3] P. D. Evans, N. Mekel-Bobrov, E. J. Vallender, R. R. Hudson, B. T. Lahn, Proc. Natl. Acad. Sci. USA , 2006, 103, 18178–18183.
[4] J. D. Wall, M. F. Hammer, Curr. Opin. Genet. Dev. , 2006, 16, 606–610.
]]>
It is unusual to think of lipids when considering the fight against bacterial infections. However, we might be grateful that some people do, because they may be on to something that looks very promising for producing new antibiotics. There is compelling evidence that a group of lipids forms a canonical system that produces the bacterium’s protective coating. Without this coating, called the cell wall, the bacterium leaks and dies. Stopping the enzymes that form the cell wall are therefore targets for treating the infection.
The research into this pathway and the chemistry of the cell wall is becoming increasingly important in fighting infection. Many existing antibiotics, such as vancomycin, that also exploit weaknesses in the cell wall, are becoming less and less effective as bacterial resistance grows. This increases the need for alternatives. The canonical pathway that produces the cell wall provides a number of targets for medical intervention that could treat the infections of a number of bacteria.
The canonical pathway is shown below (Figure). What it represents is the way in which fragments of the cell wall are build up by the activity of enzymes on lipids. The lipids are therefore being used as a sort of chemical production line. The head groups of those lipids are what become the fragments of the cell wall.
At the bottom left of the diagram, the hydrophobic component from lipid II is dephosphorylated. Then a protein (enzyme) called MraY [1,2] attaches a sugar (pink hexagon, ‘M’) that has a short peptide on it, to produce lipid I. Lipid I is turned into lipid II by another enzyme, MurG [3,4]. In this reaction, another monosaccharide is added. Lipid II is flipped from the inner face to the outer face by FtsW [5], allowing the penicillin-binding proteins (PBPs) to sew it into the developing cell wall. The hydrophobic component of lipid II can then be re-used.
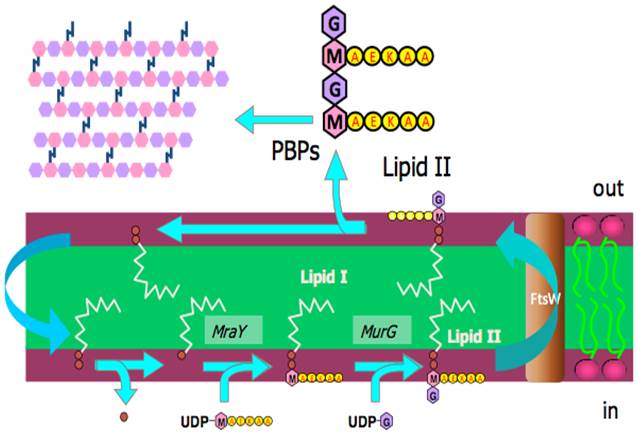
Figure. The formation of fragments of the cell wall (top left of diagram) from lipid II. Lipid II is formed in the inner face of the plasma membrane, the past step of which is performed by MurG before the lipid II is flipped to the outer face by the protein FtsW. Lipid II is made from lipid I, Lipid I is made from a recycled component of lipid II. Coloured hexagons represent monoaccharides, red discs are phosphate groups, the yellow discs marked AEKAA represent a peptide. PBP = penicillin-binding proteins. Diagram courtesy of Drs Eefjan Breukink and Tamimount Mohammadi.
Much work has gone into elucidating this pathway [1-5], in order to ensure that all the components are properly characterised and their roles understood. The understanding of these components provides a valuable set of targets for tackling bacterial infection. The activity of MraY, MurG and FtsW are all important for the formation of the pieces that make up the cell wall, and so anything that inhibits their activity has potential as an antibiotic. Watch this space!
References
[1] D. S. Boyle, W. D. Donachie, J. Bacteriol., 1998, 180, 6429-6432.
[2] B. C. Chung, J. Zhao, R. A. Gillespie, D. Y. Kwon, Z. Guan, J. Hong, P. Zhou, S.Y. Lee, Science, 2013, 341, 1012-1016.
[3] D. Mengin-Lecreulx, L. Texier, M. Rousseau, J. van Heijenoort, J. Bacteriol., 1991, 173, 4625-4636.
[4] E. van den Brink-van der Laan, J. W. P. Boots, R. E. J. Spelbrink, G. M. Kool, E. Breukink, J. A. Killian, B. de Kruijff, J. Bacteriol., 2003, 185, 3773-3779.
[5] T. Mohammadi, V. van Dam, R. Sijbrandi, T. Vernet, A. Zapun, Ahmed Bouhss, M. Diepeveen-de Bruin, M. Nguyen-Disteche, B. de Kruijff, E. Breukink, EMBO J., 2011, 30,1425–1432.
]]>
As a scientist, I am often asked to explain aspects of how science works, and why things do not work in what feels like an intuitive way from the questioner’s perspective.
A good example is a question from devout Christians about why if we call evolution a theory, we cannot also call creationism a theory. The answer is a straightforward one, but I enjoy it because it can make for a debate that goes somewhere, and is generally not one that either party comes out of feeling too bruised.
What is often difficult to explain are the subtler points of the development of theories. It is easy to give a broad-brush answer along the lines of lots of people contributing over a number of years, typically from different fields that shape a theory and continue to make it testable. However, the confusion on the faces of non-scientists makes this answer a bit useless. Something that one cannot indulge in during a social discussion is the solid use of case studies (I have yet to meet someone who can manage the tonal shift of turning a drinks reception into an impromptu PowerPoint presentation about the development of science).
One such case study about how science has developed in practice, is our understanding of a group of lipids based on a molecule called sphingosine (Figure). The chronology of this development starts in the nineteenth century with its discovery, by Johann Ludwig Wilhelm Thudichum, a pioneer of brain, and thus lipid chemistry. It is generally believed that he named it after the ancient Egyptian Sphynx because he observed that it had an enigmatic chemistry*.
What is clearer is that sphingosine is the central building block of the class of lipids known as sphingolipids. The secondary amine (nitrogen atom, N, Figure) is acylated in vivo (in living organisms) to give a sub-class of lipid called ceramides. These were named after the tissue in which they were discovered, the cerebellum, and are roughly analogous to the glyceride portion of glyceryl lipids.
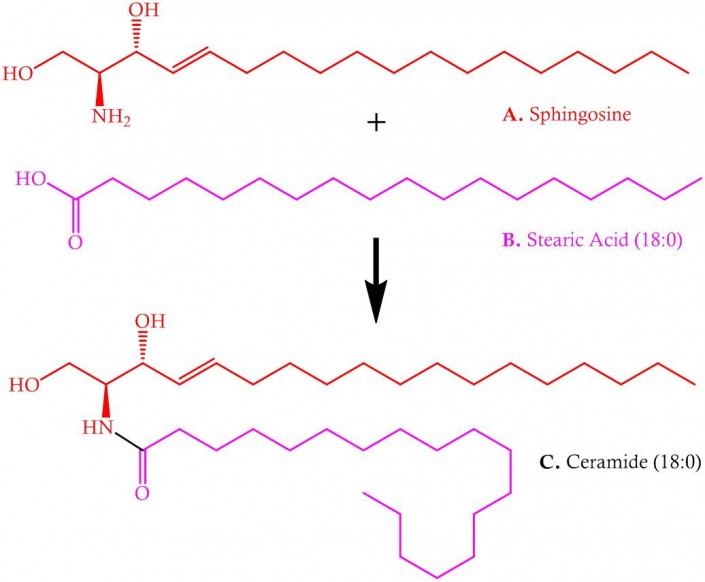
Figure. The acylation of sphingosine (A) with fatty acid (B) to give ceramide (C). The black bond in C represents the newly-formed amide bond.
Further work after their discovery increased our understanding of the lipid portion of eyes, in particular [1], establishing what sort of lipids comprised the lipid fraction (1857), and noting how this fraction differed in some diseases (1914). How the lipid fraction shifted with age took longer, with landmark discourses only well into the twentieth century (1935 and 1965).
These discoveries were made decades apart. The length of time the story of sphingolipids unfolds over is therefore perhaps partly a result of their enigmatic nature. At the time of the discovery of sphingosine, the techniques for establishing molecular structure were time-consuming, painstaking and not always conclusive. Such studies were also only possible after considerable effort had been put into purifying the analyte. It was not until the late twentieth century that biochemical techniques had developed sufficiently, and enough work had been completed, to produce a clear picture of what sphingolipids do [2] and where they are located [2-4].
Lipidomics techniques using mass spectrometry, developed only in the last decade, have shed light on the range of sphingolipids in vivo [5,6]. Spectral techniques for determining molecular structure have contributed to the demonstration of novel sphingolipids, such as 4,5-dihydrosphingomyelin [7] – the largest component of the lipid fraction of the human lens. The fatty acid component of sphingolipids alone is worthy of study, with recent work showing some of the complexity of this fraction [9].
The progress in the chemistry and biochemistry of sphingolipids coincided with an increased research effort into the physical behaviour of lipids in vivo, with recent work providing conclusive evidence about their physical properties [8].
This exciting increase in the understanding of sphingolipids, from a variety of research fields, remains strong. Some very recent research provides evidence that sphingolipids have a role in the physical process of division in at least one type of mammalian cell [5]. This physical role of this class of lipids is an unexpected contrast to the biological role sphingolipids have as molecular messengers in processes such as angiogenesis, neurodegeneration and diabetes. However it may not be unique; it appears that inositide lipids have both structural and biological roles.
Even this short explanation of the history and recent developments of sphingolipids defies understanding at first glance. It is an example of how scientific discovery follows a non-linear trajectory, suffering and benefitting from events that are unconnected with its intellectual and practical value. Such a genesis is much better described by an historical account. A straightforward and simple account of ancient ceramics, many of which date from the same time as the enigmatic Sphynx, as well as later civilisations like the Mycenaeans are good examples.
Unlike the ancients, however, the story of the sphingolipids is not yet over. We have yet to marry the dual biological and physical roles entirely, and we have yet to pinpoint the roles of the individual members of this class. Only when we do, will this enigma fall into the pages of history.
References
[1] L. Huang, V. Grami, Y. Marrero, D. Tang, M. C. Yappert, V. Rasi, D. Borchman, Invest. Ophthalmol. Vis. Sci., 2005, 35, 4333.
[2] Y. A. Hannun, L. M. Obeid, J. Biol. Chem., 2011, 286, 27855.
[3] S. Furse, unpublishd data, 2012.
[4] M. N. Islam, M. P. Jacquemot, S. Coursol, C. K. Y. Ng, New Phytologist, 2012, 193, 51.
[5] G. E. Atilla-Gokcumen, E. Muro, J. Relat-Goberna, S. Sasse, A. Bedigian, M. L. Coughlin, S. Garcia-Manyes, U. S. Eggert, Cell, 2014, http://dx.doi.org/10.1016/j.cell.2013.12.015.
[6] Rebecca L. Shaner, J. C. Allegood, H. Park, E. Wang, S. Kelly, C. A. Haynes, M. C. Sullards, A. H. Merrill, J. Lipid Res., 2009, 50, 1692.
[7] W. C. Byrdwell, D. Borchman, R. A. Porter, K. G. Taylor, M. C. Yappert. Invest. Ophthalmol. Vis. Sci., 1994, 35, 4333.
[8] K. P. Shaw, N. J. Brooks, J. A. Clarke, O. Ces, J. M. Seddon, Robert V. Law, Soft Matter, 2012, 8, 1070.
[9] N. Jimenez-Rojo, A. B. Garcia-Arribas, J. Sot, A. Alonso, F. M. Goni, Lipid Bilayers containing sphingomyelins and ceramides of varying N-acyl lengths: a glimpse into sphingolipid complexity, Biophysical Society Conference, 2014.
*However it is unclear why this particular enigma was chosen, and not one of the many others he must have been aware of (e.g. La Giaconda), some of which seem at least superficially more appropriate to a researcher of brain chemistry (e.g. Michelangelo’s ‘hidden’ anatomy lesson in the Sistine Chapel).
]]>
When I want to explain my research to someone not from a science background, I normally opt to talk about it through a fundamental process that I am sure they will have heard of. Cell division is usually the best vehicle, and it segues nicely into a discussion about what I want to do in research in the long term if the conversation goes that far.
I start by explaining that we can draw an analogy between a cell and a house. If one were an alien and wanted to study houses, the things that move and go in and out of the house would probably be the first things that would gain one’s attention; the water, electricity, people and so on. However, I suggest, if you put the materials that allowed all of those things to happen together, a house would not emerge. A pile of pipes, windows, wires and so on is the contents of a skip and not Hampton Court Palace.
In terms of the raw material, the difference between the pile of stuff and the Tudor-Baroque masterpiece I mentioned can be summed up as the bricks. This is where I draw it back to lipids: they are a lot like bricks, I say. They are the bricks of the cell. Individually, they are unexciting and almost pointless. When put together in a coherent manner, great structures can emerge—everything, in fact, from a royal residence in Surrey, to a two-up-two-down in the east Midlands.
That is a neat explanation and usually satisfying, and that does not last longer than half a glass of merlot. Sometimes their interest goes further, and so I take the opportunity to push things up a notch; I take a slightly different approach. I explain that in terms of cells, we know a lot about DNA and how it behaves when cells divide. I mention Rosalind Franklin. Then I mention that proteins have been studied extensively and that we know that they make up some important molecular machinery of the cell. I mention that Nobel prizes have been won by British scientists for work on individual proteins in this story (Sir Tim Hunt and Sir Paul Nurse). Then comes the gap in our knowledge: the cell membrane.
Despite a great deal of research effort into cell division and related processes, like vesicle budding, I say that we know little about how membranes expand, bend and divide in living cells (in vivo). There is evidence about lipid behaviour from studies of model systems, and so we are confident of the basics. We understand about different types of lipid and the different sorts of assemblies they form. I also say that we know there are an amazing number of distinct lipids but we do not know how these might change in the process of cell division.
However, breaking news sheds light on the role of lipids in living systems. Some recent work by Atilla-Gokcumen et al. [1] has indicated that the concentration of certain lipids does change during the cell cycle. This exciting work, that became available to read in the journal Cell in January 2014, reports that eleven lipids with known chemical structures accumulate in dividing cells. This set includes phosphatidylinositol, several sphingolipids, phosphatidic acid, and a phosphatidylserine as well as two pseudo-lipids (a triglyceride and a sterol derivative).
This discovery shows that lipids may have a physical role in the process of cell division. This is a significant step forward; much of the evidence to date has been limited to model systems [2,3], has been theoretical discussion [4] or a best guess based on thermodynamics [5].
One of the reasons this discovery is important is that it is not an end in itself. Like virtually all good science, it opens up a multitude of options. It invites investigation of the behaviour and interactions of those lipids that increase in concentration in the run up to cell division. It begs the question of which lipids decrease in concentration during this process, and why. It invites fresh consideration of the evidence about those lipids that have been highlighted. It also invites interest in what changes to the lipid profile occur in other mammalian cells, and even in simpler organisms (I am working on bacteria at present, for example).
We have therefore had our appetite partly satiated and partly whetted, all at once. We have learnt a bit more about the physical process of cell division, with its connections to everything from cancer to the repair of a paper cut, and more fundamentally, that a class of molecules traditionally thought unexciting, may be on the brink of celebrity.
References
[1] G. E. Atilla-Gokcumen, E. Muro, J. Relat-Goberna, S. Sasse, A. Bedigian, M. L. Coughlin, S. Garcia-Manyes, U. S. Eggert, Cell, 2014, http://dx.doi.org/10.1016/j.cell.2013.12.015.
[2] X. Mulet, R. H. Templer, R. Woscholski and O. Ces, Langmuir, 2008, 24, 8443–8447. http://pubs.acs.org/doi/full/10.1021/la801114n.
[3] S. Furse, N. J. Brooks, A. M. Seddon, R. Woscholski, R. H. Templer, E. W. Tate, P. R. J. Gaffney, O. Ces. Soft Matter, 2012, 8, 3090-3093. DOI: http://dx.doi.org/10.1039/c2sm07358g.
[4] S. Furse, S. Liddell, C. A. Ortori, H. Williams, D. C. Neylon, D. J. Scott, D. A. Barrett, D. A. Gray. , Journal of Chemical Biology, 2013, 6, 63-76. DOI: 10.1007/s12154-012-0090-1 .
[5] H. T. McMahon, J. L. Gallop; Nature, 2005, 438, 590-596.
]]>
2014 is notable for a number of centenaries. The outbreak of World War I is probably the most obvious of these, but the Nobel Prize in physics being awarded to Max von Laue for his work on X-ray diffraction is also worthy of a mention. One centenary that is almost lost to history is the publication of a paper [1] by a paediatrician called Albert Niemann, describing a disease that would later take his name. A subsequent study of the pathology of this condition was published in the 1920s [2] by Ludwig Pick. The condition is now known as Niemann-Pick disease (NPD).
The hundred years since its description has allowed much time for NPD to be tested and recorded, as well as individual cases being brought to public attention. The disease is most common in Ashkenazi Jews, who also have the highest incidence of another lipid-based genetic disorder – Tay-Sachs disease.
NPD is caused by mutation in the SMPD1 gene [3]. A number of different mutations in this gene that cause NPD have been found, several only very recently [4,5]. These mutations mean that the enzyme called sphingomyelin phosphodiesterase is not produced correctly. This enzyme breaks down a lipid called sphingomyelin (Figure) and so without a functional enzyme, this lipid builds up.
Figure. The structure of sphingomyelin. The red portion is the choline head group that this lipid shares with phosphatidylcholine. Image courtesy of Lipidmaps.
The build-up of sphingomyelins that characterises NPD occurs principally in the liver and spleen. This makes the middle-abdomen engorged and is the principle cause of death in NPD sufferers. Some treatment is available for the symptoms, and others are at the research stage, but currently no cure is available.
The severest cases lead to death before the age of two. Patients with a less severe case and ones in which treatment has been administered, have been known to live into early adulthood [3]. The universally negative prognosis for this condition invites interest from scientists, charities and news organisations alike. Scientific research, such as that conducted at a laboratory in Texas [6] is beginning to provide clues to therapeutic approaches, but despite this effort, a cure is not yet available.
Such rare genetic diseases have a clear emotional impact on a family and thus raise human interest. This also means that they often catch the attention of the mainstream press, such as an article in the Telegraph and one in the Daily Mail. The latter report handled the story with what appears to be uncharacteristic balance and restraint, both to the patient and those close to him, and to the science that underpins what we understand of the disease. It is easy to see that the human-interest factor in these stories makes them ripe for public attention by commercial newspapers. But is that the only factor that should be considered?
A more thoughtful point of view might be that the affliction of a rare genetic disease first described a century ago, on individuals not in the public eye, is at odds with the journalistic definition of news. Although this may be a fair comment, it does not say anything about the quality of the reporting of these stories. The clinical and scientific knowledge that underpins these reports has been represented reasonably well in some cases. The science has not been neglected or twisted for the sake of added drama although some coverage has used unnecessary soundbites for effect, e.g. “Childhood Alzheimers”.
The relatively good journalistic handling of the underlying causes of NPD means it has fared better than other stories with a scientific or medical background. The reporting of an observational study [7] on diet and health, left many scientists flabbergasted if not outraged. The reporting was negligent and unworthy of the organisation from which it came. However, though also flawed, the study that was being reported on was new and dealt with an issue that affects millions of people world-wide (obesity) making it newsworthy.
So, on one hand, we have reports of heart-breakingly emotional stories about individuals in which well-established science is reported soundly. On the other there is poor reporting of flawed science that may affect millions of people, albeit without tensioned heartstrings. Perhaps we need a level of human interest in any given story to ensure good reporting of science. Or perhaps the difference between childhood death from incurable genetic disease and obesity can be summed up as “one death is a tragedy, one million deaths is a statistic”?
References
[1] A. Niemann, Jahrbuch für Kinderheilkunde und physische Erziehung, 1914, 79, 1–10.
[2] L. Pick, Ergebnisse der inneren Medizin und Kinderheilkunde, 1926, 29, 519–627.
[3] M. W. Spence, J. W. Callahan. “Sphingomyelin-cholesterol lipidoses: The Niemann-Pick group of diseases” in The Metabolic Basis of Inherited Diseases, volume 2. Edited by C. R. Striver. A. I,. Beaudet, W. S. Sly, and D. Valle. Eighth edition, 1989, 0070607303.
[4] P. Irun, M. Mallén, C. Dominguez, V. Rodriguez-Sureda, L. A. Alvarez-Sala, N. Arslan, N. Bermejo, C. Guerrero, I. Perez de Soto, L. Villalón, P. Giraldo, M. Pocovi. Clinical Genetics, 2013, 84, 356-361.
[5] A. Aykut, E. Karaca, H. Onay, S. Kalkan Ucar, M. Coker, O. Cogulu, F. Ozkinay. Gene, 2013, 526, 484-486.
[6] B. Liu, S. D. Turley, D. K. Burns, A. M. Miller, J. J. Repa, J. M. Dietschy.. Proceedings of the National Academy of Sciences of The United States of America, 2008, 106, 2377-2382. doi: 10.1073/pnas.0810895106
[7] S. Rohrmann et al.. BMC Medicine. 2013, 11, 63. doi:10.1186/1741-7015-11-63
]]>The difference between you and I, and a sponge, is cell signalling. Sponges—the sort that have lived on the sea bed for many millions of years—are masses of cells that are biologically indistinguishable. You and I are made of countless different sorts of cells that between them do all the different things required for us to be walking, talking human beings.
Like a large company, all those many different cells need to be marshalled so that they work together and thus function properly. It would be no good if all cells decided they wanted to be hepatocytes (liver cells), so they must communicate with one another to ensure that the right number do the right thing at the right time.
Inevitably, this is an immensely complicated process, and not one we fully understand. Even simpler-sounding processes like dealing with sugar, or moving our muscles requires several layers of communication, or signalling, between and within cells. It was this realisation that led scientists, particularly biochemists, to look for so-called signalling molecules. Once they started looking, they found them in abundance. The process is still going on today: last month a paper was published, providing evidence that a lipid previously thought to be essentially structural in nature, may have a signalling role [1].
Signalling molecules come in a variety of sorts. Some have very familiar names, like oestrogen and testosterone. Some dissolve in the cytosol (the the watery fluid inside a cell) and in the extracellular medium (the watery fluid outside cells), where others are based in a given location, like a membrane. The inositide lipids, a group that includes PIP2, are signalling molecules that are located in membranes. Their amphiphilic nature makes this inevitable initially, but there is a twist.
Part of how the PIP2 signal is made is elaboration of the water-soluble head group of the lipid. Processing of the PIP2 signal involves separating this head group from the fatty part, leaving a molecule called IP3 and a diglyceride (DAG). IP3 is water soluble and so diffuses into the cytosol, where it meets other membranes and releases calcium. This process is required for muscles to work. It also is involved in cell proliferation (growth and division), and when it goes wrong is responsible for well-known diseases such as Huntingdon’s chorea and Alzheimer’s disease. DAG’s functions as a signal are similarly broad. It activates a group of proteins called PKC. These proteins are responsible for ‘switching on’ other proteins. As you might imagine, the down-stream effects of this are several, including muscular activity, but also hormone secretion, and the production and metabolisation, of fats.
This understanding is the result of about 50 years’ work by biochemists. One might think that that would be enough, but scientists have other ideas. Interest from scientists who research the physical behaviour of lipids has prompted interest in the inositide family [2,3], which of course includes PIP2. Several biological studies have shown that PIP2 is present during important cellular events, such as the formation of vesicles [4] (part of the process of carrying nerve impulses) and during one called endocytosis [5,6,7], which is similar to what immune cells do to kill bacterial cells. Some evidence about the physical behaviour of inositides has already been published [2,3], much of which was surprising and so invites questions about what the others, like PIP2, do.
The chemistry of PIP2 is remarkable with respect to other lipids. Its large head group has complicated interactions with water and acids [8], that probably influence its physical behaviour and its molecular interactions with proteins, and thus its biological behaviour.
‘Probably’ is a very dangerous word in science, unfortunately. It frightens us scientists, making us want to reach for evidence and to run away from anyone who does not. Despite that, there is a solution. And it has a similarity with PIP2 itself. The answer is to research this molecule using more than one scientific discipline, together. So, like the PIP2 molecule with its several effects and influences, we need scientists who can do several different things.
This is not a new idea. In the nineteenth century, the science of ‘bio-chemistry’ was born, when it was realised that there was a good deal of chemistry in cells, and that understanding that chemistry was key to understanding how cells work. In the twentieth century, research questions such as the structure of DNA were solved by using physics and physical techniques. This is the basis for biophysics. Another form of this, medical biophysics, is useful for researching physical processes in disease. For example, the reason why the heart pumps blood is because of a change in pressure in the chambers of the heart. When that goes wrong, it is a physical process as well as a biological one, and often, so is the solution.
So, how does this fit with PIP2? The multi-disciplinary work that is tackling that problem is called chemical biology. This term refers to the use of chemical and physical techniques to solve research questions in biological systems. This means it includes using LASERs to treat arthritis, computers to model protein behaviour, but also x-rays to work out what lipids do. This sounds very general, and it is. Focus is important in science, but in chemical biology the focus is on the problem, rather than how it is solved. The point is to identify the question and then use whatever techniques, from whichever discipline, are required to answer it thoroughly. This is what holds the key to weaving the disparate threads of PIP2, of arthritis, of protein behaviour and all sorts of other things, into a coherent understanding.
References
[1] S. Liu, J. D. Brown, K. J. Stanya, E. Homan, M. Leidl, K. Inouye, P. Bhargava, M. R. Gangl, L. Dai, B. Hatano, G. S. Hotamisligil, A. Saghatelian, J. Plutzky, C. H. Lee, Nature, 2013, 502, 550. DOI: 10.1038/nature12710
[2] S. Furse, N. J. Brooks, A. M. Seddon, Rudiger Woscholski, R. H. Templer, E. W. Tate, P. R. J. Gaffney and O. Ces, Soft matter, 2012, 8, 3090-3093. doi: 10.1039/c2sm07358g
[3] X. Mulet, R. H. Templer, R. Woscholski and O. Ces, Langmuir, 2008, 24, 8443–8447. doi: 10.1021/la801114n
[4] I. Milosevic, S. Giovedi, X. Lou, A. Raimondi, C. Collesi, H. Shen, S. Paradise, E. O’Toole, S. Ferguson, O. Cremona and P. De Camilli, Neuron, 2011, 72, 587-601.
[5] R. Zoncu, R. M. Perera, R. Sebastian, F. Nakatsu, H. Chen, T. Balla, G. Ayala, D. Toomre and P. V. De Camilli, Proceedings of the National Academy of Sciences, 2007, 104, 3793-3798.
[6] G. Di Paolo and P. De Camilli, Nature, 2006, 443, 651-657.
[7] P. DeCamilli, S. D. Emr, P. S. McPherson and P. Novick, Science, 1996, 271, 1533-1539.
[8] E. E. Kooijman, K. E. King, M. Gangoda, A. Gericke; Ionization Properties of Phosphatidylinositol Polyphosphates in Mixed Model Membranes, Biochemistry, 2009, 48, 9360-9371.
]]>
The broadest definition of lipids includes steroid hormones like testosterone and oestrogen. These types of molecule do not have self-assembly properties and so are not classical lipids. This does not detract from the research interest they generate however. Sex hormones and their equivalents are a powerful initiator, indicator and tool in animal systems. This is simply because sexual maturity, and thus reproduction, is impossible without them.
It is therefore easy to understand the interest in molecules that have some of the characteristics of sex hormones. These might find a use to induce the part or all of the response of sex hormones for certain types of hormonal chemotherapy. They might provide tools for understanding the complicated sets of biological responses to such hormones in model systems, in order to better understand processes such as puberty.

Figure 1. Potamopyrgus antipodarum (New Zealand mus snail), picture courtesy of Wiki commons, http://en.wikipedia.org/wiki/File:New_Zealand_Mud_snails.jpg.
What might garner a more sensational focus, would be where such molecules appear where they should not, and thus where they can cause damage. Some evidence for this was a study published in 2009 in which a set ofPotamopyrgus antipodarum, the New Zealand mud snail, were grown in glass and plastic bottles [1]. What the authors, Wagner and Öhlmann (also spelt Oehlmann) report, was that the snails grown in plastic bottles showed a greater endocrine response than those grown in glass bottles. The plastic in question was poly (ethyl terephthalate), often known as PET, a plastic commonly used to make bottles for drinking water, fruit juices and so on. This plastic typically contains traces of bisphenol A.
The link between the action of bisphenol A and hormone-like activity has been known since 1998 [2], but the link with food is enough to get both media and scientific attention. Some statistics that correlate with this: in the four years before Wagner and Öhlmann’s paper, about 1,300 articles were published on this compound. In the four years since, nearly three times that number have been published*. The paper itself has been cited 76 times, about thirty times more than the average paper in the journal in which it was published, Environmental Science and Pollution Research [http://www.springer.com/environment/journal/11356]. The research paper detailing the activity of bisphenol A at oestrogen receptors [2] has received 195 citations, nearly fifty times more than a typical paper in Molecular and Cellular Endocrinology [http://www.journals.elsevier.com/molecular-and-cellular-endocrinology/].
It is easy to criticise the sensational aspect of this work and ascribe the response as a ‘panic’ one. While this is probably, objectively, at least partly correct, there is no doubt the subsequent out-pouring of activity since has proved fruitful. Many research projects have been focussed on the compound directly. Research published last month paves the way for detection of bisphenol A in water, even at very low concentrations [3], finds a use for bisphenol A in another plastic coating [4] and updates our understanding of how and where bisphenol A is found in human metabolism, for example, in the urine of pregnant women in Australia [5].
Observational studies, such as that mentioned above [5], back up the human consumption of bisphenol A. This should not be over-stated however; Callan et al. report measuring it but no tests were carried out in this study**. This observation increases in importance when looked at in the light of another recently published study, by Machtinger et al. [6].
In this work, the team of scientists investigated the question “Does exposure to Bisphenol A affect the maturation of human oocytes in vitro?” Oocytes are the cells that become ova, the female sex cells. They transform from oocytes to ova during the menstrual cycle, in a way that is initiated and controlled by sex hormones. The conclusions of this paper therefore are striking nut not necessarily surprising: both the number and quality of oocytes was inversely proportional to bisphenol A concentration. In other words, the greater concentration of bisphenol A, the more damaging the effect. This is described by a handy piece of terminology, used by pharmacologists ‘dose-response relationship’.
The effects of bisphenol A are several though, the authors state that cell cycle progression, spindle architecture and chromosome organization during oocyte maturation are all affected by the presence of this compound [6]. It appears this will affect female foetuses in a measurable manner, as this is where oocytes are made.
To an informed scientist, this raises at least as many questions as it answers. For example, what is the effect on male foetal and sexual development? What is a safe dose of bisphenol A? Is research that uses bisphenol A in plastic coatings [4] made redundant by this work?
The fact that we are in a position to ask these questions is as a result of important steps forward in our understanding of the biological activity of this lipid analogue. The data from the study by Machtinger et al., though not a pleasing answer, has allowed us to know much more about the precise nature of the effect of bisphenol A, and should be potent enough to quench any hysteria that may have arisen, about the original observation of bisphenol A as an analogue of oestrogen.
There is no doubt, however, that the potential impact on human health was a motivating force in this work, and strengthened the case(s) for funding it. So, what science really needs is a moderate level of panic in the outside world to advance.
References
[1] M. Wagner, J. Oehlmann. Environmental Science And Pollution Research , 2009, 16, 278-286. DOI: 10.1007/s11356-009-0107-7
[2] J. C. Gould, L. S. Leonard, S. C. Maness, B. L. Wagner, K. Conner, T. Zacharewski, S. Safe, D. P. McDonnell, K. W. Gaido, Molecular and Cellular Endocrinology, 1998, 142, 203-214. DOI:10.1016/S0303-7207(98)00084-7.
[3] Z. L. Mei, W. Qu, Y. Deng, H. Q. Chu, J. X. Cao, F. Xue, L. Zheng, H. S. El-Nezamic, Y. C. Wu, W. Chen, Biosensors & Bioelectronics, 2013, 49, 457-461. DOI: 10.1016/j.bios.2013.06.006.
[4] A. M. Tomuta, X. Ramis, X. Fernandez-Francos, F. Ferrando, A. Serra, Progress in Organic Coatings, 2013, 76, 16161624. DOI: 10.1016/j.porgcoat.2013.07.010.
[5] A. C. Callan, A. L. Hinwood, A. Heffernan, G. Eaglesham, J. Mueller, J. O. Odland, International Journal of Hygiene and Environmental Health, 2013, 216, 641-4. DOI: 0.1016/j.ijheh.2012.10.002
[6] R. Machtinger, C. M. H. Combelles, S. A. Missmer, K. F. Correia, P. Williams, R. Hauser, C. Racowsky, Human reproduction, 2013, 28, 2735-45. DOI: 10.1093/humrep/det312
*Records from Thomson-Reuters Web of Knowledge, www.wok.mimas.ac.uk.
**These authors also choose to cite results that are not statistically significant, in the Abstract of the paper. In other words, the results being highlighted were not meaningful in themselves. However, this does not necessarily influence the reliability of the principle observation).
The oil we use in cooking every day typically comes from plant seeds. Olive, sunflower, rape and palm are common examples, with sesame, coconut, peanut, walnut, cotton and soya also available, though less well known. (Triffid oil is, of course, fictitious.). This array of plants that can store oil easily and in large enough quantities to be commercially viable raises the question of how this happens.
The question becomes more pertinent when we remember that oil and water do not mix. We can shake olive oil and water together in a closed, colourless container and, for a short time, observe what looks like a mush, but the oil and water separate after only a few minutes. We know plant material contains water (up to 96%); we also know that the oil is there too. The seeds in which the oil is stored are small and we cannot see the form the oil takes, suggesting the oil is in microscopic portions.
Work using light microscopes reveals this to be the case. Using staining techniques, the bodies in which the oil is stored are visible under a light microscope. This tells us about the compartmentalisation of oil storage in plant seeds, but not how it seems to be energetically favourable*: it looks for all the world that the oil is in blobs in a watery system (a cell). It looks like what food technologists call an oil-in-water emulsion: small blobs of oil suspended in water.
This on its own is impossible for any length of time, as we observe. There must be a third component that satisfies the energetic requirements of the apparent observation of oil in water. That third component is a mono-layer of amphiphilic molecules. Like simple lipids, this mono-layer comprises a surface that is hydrophilic (likes water) and one that is lipophilic (likes fat).
This is an elegant compromise between the requirements of storing oil in a seed. However, it raises as many questions as it answers: how does the plant control this important organelle? What is the mono-layer made of exactly? Do the components of this mono-layer have other uses?
The answers to these questions share a few connections. We know from a recent study detailing the molecular composition of this organelle [1], that it is dominated by a group of protein isoforms known collectively as oleosins. These proteins are amphiphilic, but also have other properties.
For example, many seeds that use oil as an energy storage desiccate on maturing, i.e., lose some of their water. We know that lipid systems are sensitive to water, and so when the amount changes, it can have a profound effect on lipid behaviour. However the oil bodies, the organelles that store the oil, remain in tact. This is clearly an important and transferrable feature: all oil bodies yet discovered comprise large amounts of oleosin. It turns out that the oleosin component is not only an amphiphile, but one that can confer a measure of flexibility on the mono-layer it comprises. The oleosin fraction of the surface is typically the dominant one; about 60% of the mass of the oil body mono-layer in Helianthus annuus (common sunflower) was found to be oleosins [1].
This suggests that this protein may have a powerful part to play in other applications – and there are a surprising number. In fact, anything with an oil-in-water emulsion that is in any way sensitive, may be improved by the use of oleosins. Current speculation and applications under investigation include a molecular vaccine delivery mechanism (increasing the stability of the vaccinating molecular species) [2], a way of immobilising enzymes in a way that is recyclable [3-5] and a way of improving the shelf-life of edible oil-in-water emulsions [6-9].
In fact, the list goes further than these examples, and is fast approaching the magnitude of plants that make oil bodies in quantity. The scope for a flexible mixture of lipidic and proteinaceous amphiphiles is therefore something keenly anticipated, and not just by lipid chemists.
References and Notes
[1] Furse et al.,
[2] H. Deckers et al.,
[3] C. J. Chiang, C. Y. P. Chen, J. T. C. Tzen,
[4] J. H. Liu, L. B. Selinger, K. J. Cheng, K. A. Beauchemin, M. M. Moloney.
[5] J. R. Liu, et al.,
[6] I. Fisk, R. Linforth, A. Taylor, D. A. Gray,
[7] I. Fisk, D. A. White, M. Lad, D. A. Gray,
[8] D. R. McCaskill, F. Zhang,
[9] C. V. Nikiforidis, V. Kiosseoglou.
*This typically means the conformation of the system that requires the least energy. It is also used to describe a conformation of the system that does not require more energy than the system possesses.
Here is a dangerous opinion: murderers should not be killed.
It is an opinion you have probably heard before, and if the statistics I have heard about are right, and you live in western Europe, the chances are you agree with it.
However, in India, they do not. At least, they do not agree with it to the point of changing their statutes to reflect this. We have seen a prime example of this, this week, with the announcement that the four men convicted of raping and causing the death (murder or manslaughter is not clear) of a woman student, have been given the death penalty. Another man who was alleged to have participated in the incident has already died (there is evidence he hanged himself, though his family deny this) and one male who was 17 at the time has been sent to a juvenile reform institution.
This case therefore provides food for thought for those who oppose the death penalty but who also have a strong interest in tougher, and greater, sentencing of rapists. In the UK, current attitude suggests that rape is a more sinister and politically incorrect crime than murder or manslaughter, so one might think that a death penalty should be awarded for that crime, if any, especially if it is accompanied by the death of the rape victim. It seems that international organisations are also starting to take this view. For example, Meenakshi Ganguly, the South Asia Director of Human Rights Watch, said the treatment of individual women was “much, much more important” than whether or not the perpetrators were put to death. It is not clear under which context these remarks were made, but from the outside it starts to sound that human rights can be picked and chosen.
What interests me more than the human rights angle is India’s use of the death penalty at all. Ghandi said, referring to violent conflict, “An eye for an eye, and the whole world goes blind”. Barely more than sixty years after independence, this appears not to apply within India. Add that this particular case is unbalanced in this respect; justice is apparently served to one manslaughter/murder and six rapes by five deaths and a remedial sentence. This starts to sound more like a state-mediated revenge for the crimes at the behest of public pressure, and less like justice.
Militant feminists reading this will no doubt be angered by that view, but I am not alone in holding it. Tara Rao, Director of Amnesty International India seems to agree. He said that “Sending these four men to the gallows will accomplish nothing except short-term revenge”.
There is no doubt, for me at least, that nothing can justify the actions of rape and murder of any person. However, in the interests of equitability, we must ask ourselves whether death and a painfully unbalanced approach solve any of the problems that come under the spotlight in this case. The upshot of this is that men and women are as separated as much as they ever were, justice has compromised yet again by ignorant and political intervention and the causes and consequences of rape and murder are more unclear than ever before.
What we lack is an even-handed, cool-headed, objective approach. When are we going to get that?
I thought of another one of those euphemistic phrases they use in obituaries, last week. You know the ones – ‘he did not always uphold the highest ethical standards of the city’ means he was a thief, ‘she was a tireless campaigner for free speech’, means she was a crashing bore, ‘he did not always see eye-to-eye with the prevailing political orthodoxy of the time’, means he was a fascist. The pleonasm I thought of was for an ex-colleague of mine, an academic of sorts: ‘S/he had an unorthodox approach to authorship’. Authorship, in this case, refers to the process of apportioning credit for work done for scientific publications – journal articles, patents and books.
The Structure of Turnip Yellow Mosaic Virus: X-Ray Diffraction Studies, Aaron Klug, J. T. Finch, Rosalind E. Franklin.
In case you are not familiar with it, typically, the first author is the Ph.D. or Master’s student who does the lab work and who writes the first draft of the paper. The last author is the supervisor and the person who wrote the grant proposal that funded the project. The second author is perhaps the post-doc who re-wrote the paper or the one who was the go-to person in the lab when the student needed to learn a new technique or problem-solve. The second-to-last author is often a second supervisor, who also helped write the grant and helped out with the supervision occasionally. Mid-author positions are people who made the smallest contributions, they may have acquired and analysed some data that was included in the paper but does not make-or-break it.
I must admit I thought of the euphuism ‘unorthodox approach to authorship’ only when I was on the negative receiving end of it; I was not included in the list of authors of a publication, where I should have had a reasonably prominent position. This is not as upsetting or as frustrating as it may sound, it just means I have one fewer publication to add to my list. In any case, it is far from the only example as virtually every academic I know has been left off a list of authors unfairly at some point. Some have even written about it and the whole process, including Sylvia McLain.
In this particular case, the euphemism goes a bit further than covering up for someone’s political correctness. The ex-colleague in question also likes to take first authorship in a paper every couple of years, without having done any lab work or writing much of the manuscript. This person also likes to include people (sorry, “authors”), who have made no contribution at all to a given publication, as a way of shoring up their perhaps weak, position. So, it may be an hyperbole of sorts, but it is not too unfair itself.
The argument against this gripe I raise is that these things are really convention. There is no set rule, they differ between disciplines (authorship in engineering and science are in the reverse order with respect to one another), and there is no one to arbitrate claims of it going wrong. Authorship therefore has a political and even subjective slant—and so getting what you deserve really requires more effort that just being in the lab and working hard. You must be memorable to the person who presses the submit button that sends the manuscript to whoever publishes it.
This political element to publishing science leaves everyone open to equally subjective judgement about the decisions made, or the names forgotten. In other words, I am aware that someone may therefore think of an obituary euphemism for me at some point. I wonder what it might be. In a literal sense and by my own admission, I do not suffer fools gladly. I do not like having my time wasted, less still by people who are stupid or have no idea they have done it. According to Stephen Fry, where this pleonasm is used, the real meaning is that this person is “a howling shit” (on Youtube, from 10’40”). I hope I am not that, but I cannot deny that it is not a million miles away from finding stupid people boring.
Recently, the acclaimed former ballerina Darcey Bussell was quoted in a national newspaper extoling the virtues of “Sanctuary Spa Lipid Recovery Facial Oil”. The product name given on the website of a national pharmacist is slightly different, “Sanctuary Active Reverse Lipid Recovery Facial Oil” but appears to be the same product. A product with that name is also available on at least one on-line auction website and through other retailers in the UK and abroad.
The name used for this product interested me for several reasons. The first, of course, was that it contained the word “lipid”. The second was that the name also contains the terms “active reverse” and “recovery facial”, and because I am not sure I understand them. Taken at face (!) value, I imagine they are broadly along the same lines as “anti-ageing”, but they seem less clear than that term. The cynic in me immediately asserts that this is because the term ‘anti-ageing’ has been discredited: demonstrable evidence of a reversal of the process of ageing by applying oil-in-water emulsions to the surface of the skin, has yet to reach the public domain. However, the product label does contain some more text that allows us to deepen our understanding of what the manufacturers suggest it is for, and how it should be used:
“Lipid complex: re-charges, hydrates & protects cells from ageing to provide a host of visible benefits on the skin’s surface.”
The term ‘Lipid complex’ has a particular meaning in chemistry, that is not in common usage, nor is relevant to the ingredients listed on the product (see below). It is not clear to me what “re-charging” is in this context, only that it sounds like the sort of thing that one might want, whether or not it makes clear sense. “Hydrates” on the other hand, is clearer, is means to expose to water, especially in a way that moistens a surface or mass at a molecular level. Hydration of skin can of course be achieved with water itself, as anyone who has had a long bath will know. However, the list of ingredients do not include water, so hydration is apparently not possible—unless it is contained in one of two accompanying products, “Peptide Protect Day Cream SPF20” or “Peptide Replenish Night Cream” that are associated with it. However, they would be the hydrating agents, and not this preparation.
The next phrase “protects cells from ageing” is more dangerous, and I am afraid to say, an out-and-out lie. The cells on the surface of our skin are dead and so are not involved in the ageing process. So, simple logic tells us that there is no way that they can be protected from it. This phrase also sounds like something deliberately designed to be unclear but that sounds vaguely like what one might want.
These observations make one wonder what this preparation will do. We can start by looking at the ingredients:
“Helianthus annuus (Sunfower) seed oil, Simmondsia chinensis (Jojoba) seed oil, Triticum vulgare (Wheat) germ oil, Rosa rubiginosa seed oil, Caprilic/capric triglyceride, Lavandula angustifolia (Lavender) oil, Linalool, Punica granatum seed oil, Geranylgenarylisopropanol, Anthemis nobilis flower oil, Citrus aurantium amara (Bitter orange) flower oil, Limonene, Geraniol.”
The first four ingredients, and thus the bulk of the material in this preparation, are all oils. “Punica granatum seed oil” is the oil from pomegranate seeds. In chemical terms, these are triglycerides [link] that are a liquid at room temperature. Chemically they are very similar to olive oil and so the effect of rubbing that on your skin will be very similar to this preparation. What this preparation contains that olive oil does not, are various perfumes: lavender oil, linalool, Anthemis nobilis flower oil (better known as chamomile), Citrus aurantium amara (Bitter orange) flower oil, Limonene, Geraniol.
There is no doubt that this mixture would provide a sort of aesthetic experience that may improve mood, however, none of these ingredients has been shown to be able to reverse the effects of activity, the latter being the apparent claim of the product name.
This leaves us with one ingredient, “Geranylgeranylisopropanol”. This is a non-standard name for one of a collection of organic compounds produced mainly by plants, called terpenes. This ingredient has been included in at least one patent related to the treatment of medical disorders and is “rated” by writers of guides to cosmetic ingredients. However, according to the Thomson-Reuters database of scientific research (wok.mimas.ac.uk), no research has been published about this compound. It is therefore unclear what it may or may not do. The structure it has (at least according to a patent, that lists it) suggests that it probably has amphiphilic character, so could be classified as a lipid. It is unlikely to self-assemble as the polar head group is too weak, but it is probably amphiphilic nevertheless.
This leaves the current product investigation facing a damning conclusion: this product is unlikely to be what shoppers think it is, nor to have the effect that the deliberately-unclear name suggests. At best, there is no evidence for the claims apparently being made. Despite these, the product has not been exposed as a scandal (unless this article is the start, but I doubt it), it is still sold widely and it has not been withdrawn or re-modelled as a result of the application of the Trades Descriptions Act. There is some suggestion that the product contains an amphiphilic species, a lipid, so that part of the name appears to be correct.
The question in my mind now is: what is the point of this product? It is not flagrantly unlawful, but it serves no obvious purpose either, other than it might smell nice. Is this product part of a conspiracy to test Geranylgeranylisopropanol on a large group, an unsuspecting public?
]]>
Sieves are excellent tools for separating particulate matter based on size. It is how flour is separated from chaff in milling (wire mesh) and how Mary Berry sifts flour when baking a cake, it is how water is separated from cooked pasta (colander) and it is part of the process of preparing sand for building. It therefore seems likely that at least once a day something is filtered on our behalf, and that we therefore probably all filter something or other at least once each week for ourselves.
Cellular membranes and the membranes of the compartments (organelles) within cells are excellent filters. They need to be; waste must be lost and nutrients from the surrounding medium must be taken up. Additionally, the correct water balance (osmotic pressure) must be maintained, something that requires the cell to be able to move water in either direction. This is a lot of work for a something that is only two molecules thick.
Lipid membranes are therefore sophisticated filters that put the preparation of cooking ingredients distinctly in the shade. However, with this range of jobs comes a degree of vulnerability. The permeability of membranes is combatted with careful control mechanisms: it is not useful to the cell to take on more water than is necessary, or fail to take up nutrients such as metal ions, that it needs. In the case of metal ions, the control mechanism is particularly well developed, but also elegantly simple.
Metal ions typically have one or two positive charges. Group I elements like sodium and potassium have a single charge, Group II elements like calcium and magnesium have two positive charges. Such positive charges are electrically attracted to negatively charged species – something at the basis of the observation that opposites attract.
But what would happen if we had a permeable membrane between a medium containing negatively charged ions and one containing positively charged ions? There would be a strong, electrical desire for ions to move across that membrane. This attraction between ions of dissimilar charges across a membrane is known as an electrical gradient. There is a high concentration of positive charges on one side that is attracted to a high concentration of negative charges on the other side. But what if this movement of ions is not useful to the cell? What if it needs to retain a high concentration of positive ions within the cell and prevent the entry of negatively charged ions from the medium? Normally, this function is performed by membrane proteins called ion channels that have evolved to transport ions through a pore. This process requires energy, but it provides very fine control of the movement of ions. These channels are useful as they mean that the cell has control over the movement of these ions.
Some times however, a bulk movement of ions is required – for example in the movement of a neuronal impulse. In this case, the fine control of some types of ion channel is not quite as necessary. For this purpose, a mechanism that does not even require a protein has been exploited by cells. This mechanism occurs through voltage gated lipid channels. As the name suggests, the movement of ions through such channels is controlled by voltage. If the potential difference (measured in volts, V) is sufficiently high, the gate (pore) will open and the ions are lost to the far side of the membrane. Recent work by Blicher and Heimburg [1] has shown that this is comparable to that of protein channels.
In light of the efficacy of these lipid channels, you may wonder why protein ion channels ever evolved. It has been tentatively suggested (Blicher and Heimburgh [1]), that differing pH and temperature influence the size and behaviour of lipid pores, rendering them somewhat inconsistent. Protein channels are more dependable and provide a greater consistenty, allowing the cell to retain control of ion movement irrespective of changing conditions. However, this has yet to be tested formally.
References
[1] A. Blicher, T. Heimburg, Voltage-Gated Lipid Ion Channels. PLoS ONE, 2013, 8, e65707. doi:10.1371/journal.pone.0065707
]]>Unsaturated fats are often regarded as healthier than their saturated equivalents. This translates into eating fish rather than beef burgers, chicken rather than pork and olive oil rather than butter or lard. These sorts of dietary changes have calorific advantages for those wishing to reduce their dietary intake of energy, as unsaturated fats contain slightly less energy than saturated fats.
Unsaturated fats are often described as having a number of health benefits, including reports in the popular press that they may ease type II diabetes and reduce heart disease. Although much of this evidence is not yet conclusive for the general population – with anecdotes about some sources of saturated fat sounding counter-intuitive at first sight – we do know a lot about the basic chemistry of unsaturated fat.
Anyone who has cooked with unsaturated fat (cooking oil) and left it exposed to the air for a period afterwards will be aware that the fat will become rancid – it rancidifies. Rancidification is a reaction between the unsaturated bonds in the oil and oxygen from the air; the unsaturated bonds are oxidised. This is what makes the flavour and smell of the fat much less attractive.
Unfortunately, this sensitivity to oxidation is inherent in unsaturated fats. In fact, the more unsaturated, the easier it is for them to rancidify. A further problem is that such fats do not need to be exposed to the air for oxidation to occur. Oxidation can also happen to fats in our bodies. Oxidation of fats in our cells happens much more slowly than in air, and happens when the fatty acid residues are attacked by oxygen radicals.
This phenomenon relating to the fatty acid residues in lipids in the eye has been discussed in this blog already, but there is mounting evidence that suggests oxidation of fatty acids and the metabolic products are important for normal metabolic function, but are also important in dysfunctional systems, including in the brain [1], in atherosclerosis [2] and in tissue repair [3].
If such a process is so widespread and influential, you may wonder why it has taken so long to come to light. The good news is that most life forms have evolved to be able to metabolise the oxidised fatty acids, removing them from the system. Recent research by Teixeira et al., [1] suggests that one method involves dietary intake of a specialised kind of fatty acid. This fatty acid contains what is called a furan ring (a five-membered ring comprising one oxygen atom, Fig. 1), but is otherwise not unusual.
Fig.1, Top, Furan-containing fatty acid, known as F6; Bottom, eicosodienoic acid, an unmodified fatty acid that may be a precursor to F6.
Evidence from this study [1] suggests that once this modified fatty acid is in membranes, it is able to react with damaging oxygen species in the place of other fatty acids. This greater reactivity of the F6 is ascribed to its ability to ‘scavenge’ for the reactive oxygen species that would otherwise react with unsaturated fatty acids.
This mechanism is based on a replaceable molecule that suffers oxidative damage more readily than ordinary fatty acids. It therefore protects the others by acting as a sort of ‘fall guy’. How this mechanism operates allows us to draw a number of conclusions and ideas for future work. First, modified fatty acids such as F6 are present in fish oils and so dietary intake of seafood seems a good way of improving the availability of such molecular species. Second, an idea arising from the mechanism described by Teixeira et al., [1] is that it may be possible to prevent or reduce oxidation using chemical intervention. In other words, using this mechanism as a basis, it may be possible to develop treatments for conditions that result from oxidative stress. This has implications for how conditions such as Alzheimer’s and cardiovascular disease may be overcome.
References
[1] A. Teixeira, R. C. Cox, M. R. Egmond, Food and Function, 2013, in press, DOI: 10.1039/c3fo60094g.
[2] G. Spiteller, Ann. N.Y. Acad. Sci., 2005, 1043, 355. http://onlinelibrary.wiley.com/doi/10.1196/annals.1333.042/pdf
[3] E. Novo, M. Parola. Fibrogenesis & Tissue Repair, 2008, 1, 5. http://www.fibrogenesis.com/content/pdf/1755-1536-1-5.pdf
]]>
Recently, I have had a few requests to write a post about glycerides. It really is a good idea as these molecules have important jobs to do, but are also a building material for several other lipids. In fact, the majority of lipids and fats are either glycerides, or can be made from them. What I am going to do here is go through a few basics and then give an example of what they can do.
The first thing to know is that glycerides come in three different sorts: monoglycerides, diglycerides and triglycerides (Fig. 1). These are also sometimes called mono- di- and tri-acylglycerols, respectively.
The most common glycerides are probably triglycerides, so I will discuss those first. Triglycerides are fats or oils* but not lipids, as they are not amphiphilic. Be warned, though: plenty of people lump them in with lipids anyway. However, for the avoidance of doubt I shall continue to refer to them as fats if they are saturated (like the ones from butter or lard), or oils if they are unsaturated (like the ones from olive or sunflower oil). Whatever you call them, and however they are classified, one thing is not in doubt: triglycerides are the most concentrated way terrestrial life-forms have of storing energy. This energy store can be quantified, it stores 9 kcal/g. Energy can be stored as protein and carbohydrate, but this is never quite as concentrated as fats. Triglycerides have been found in pretty much every type of living organism yet discovered – certainly in plants, insects, fungi and animals. There is less need for bacterial and archaea species to store energy as higher life-forms do, so the quantities in evidence there are much lower.
Monoglycerides are the least common of the three types in biological systems, and are often intermediates in the metabolism of triglycerides. They have some peculiar physical properties that have been explored in a number of research papers (others include this one and this one). What that work has shown, is that the sorts of lipids found in biological systems can adopt certain morphologies; in a sense, it has shown us what sorts of shapes and contortions are ‘possible’ with these types of lipid. This has implications for what we observe in the physical process of cell division, as data like this can allow us to identify the physical behaviour of individual lipids. This allows us to start the process of ‘who is doing what’ on a molecular level, during processes such as cell division.
Diglycerides are perhaps the most interesting because they exist in nature as shown above and are also a building block for many other biological lipids. They are amphiphilic by virtue of the hydroxyl group (polar end) and the two fatty acid residues (lipophilic end). However, the polar end is not quite polar enough to make diglycerides self-assemble on contact with water, unless they are mixed with other lipids, like phosphatidylcholine (PC). This places diglycerides in a peculiar sort of physical purgatory, where they are not really lipids in the classical sense, but are not fats either. Cholesterol also falls into this category as it too forms part of a biological membrane, but will not form a membrane in its own.
Adding a simple phosphate group to a diglyceride turns it in to phosphatidic acid (PA). This will self-assemble on its own on contact with water, and is the simplest common phospholipid. Further chemical elaboration of PA gives rise to the majority of biological amphiphiles. One particularly interesting one I like is called PIP2. The full name for this particular lipid is phosphatidylinositol-4,5-bisphosphate. You can see that the name is built up from ‘phosphatidyl’ (referring to the PA part), ‘inositol’ because it contains an inositol structure, called an inositol moiety, and ‘bisphosphate’ because it contains two phosphates not joined to the glyceride. This lipid is special because it is not just part of the membrane, but also a signalling lipid. We know this for several reasons, one of which is that when it is hydrolysed (cut in half), it gives rise to two molecules that go on to have other functions.
PIP2 is hydrolysed into IP3 and a diglyceride. In cells, IP3 is the trigger for the release of calcium ions from intracellular storage. The effects of this are as varied as muscle contraction and some of the early events of fertilisation. Cell type is therefore important in knowing what this signal does.
The diglyceride activates an enzyme called PKC. This enzyme phosphorylates a number of other proteins, ensuring that they are ‘switched on’. This leads to a number of carefully controlled effects, including smooth muscle contraction in the digestive system, muscle contraction that causes ejaculation and the secretion of saliva, and aggregation of blood platelet cells. PKC also activates the proteins that are involved in neuronal activity in the brain.
This variety of bodily responses means that tight control mechanisms are required at a cellular level in order that the incorrect effect is not initiated**. The thing that strikes me about this is that the effects we observe of both the signalling molecules the come from PIP2, and the different glycerides, show just how closely our cells can control all of these molecular species – enough for us not to have any conscious idea that digestive, ejaculatory and neuronal effects are triggered by the same chemical process. Further, that such similar molecules can be both an energy storage facility and a means of digesting our food.
* The difference between fats and oils is that at room temperature, oils are liquids and fats are solids. This is related to the number of unsaturated bonds in the fatty acid residues.
** Some current research is focussed on what happens when such signalling goes wrong, and on how to treat this medically.
Infectious diseases are an on-going concern in current medicine, with striking reports about viral infection in hospitals and concerns surrounding the threat of resistance to antibiotics.
Viral infection is powerful, as indicated by 1•8 million people dying every year of HIV/AIDS and 0•5 million per (non-pandemic) year from Influenza. About 34 million people have HIV/AIDS at present, with around 4 million people suffering from influenza each year.
These scales of viral infection and death have generated a lot of medical and scientific interest. One area of exploration is the structure of the virions (also known as virons), the individual virus ‘particles’, a bit like the cells that make up a species of bacterium. Our current understanding of virion structure comes from a number of sources, and was started mainly by the work of scientists like Rosalind Franklin [1] and D. L. D. Caspar [2]. Their work showed that a limited range of proteins of a consistent shape fitted together to make a discrete virion such as the tobacco mosaic virus (Fig. 1). In these assemblies, a central core of DNA or RNA is protected and transported by a protein coat. This protein coat, called a capsid is made of individual protein blocks called capsomers.
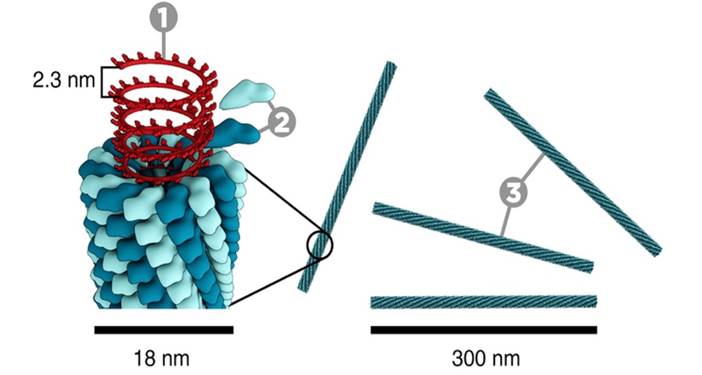
Figure. 1. The construction of the Tobacco Mosaic Virus (TMV). 1, DNA; 2, Coat proteins (‘capsomer’); 3, capsid. Photograph courtesy of www.microbiologymadeeasy.com.
More recently, it has become clear that some viruses have evolved to include a lipid mono- or bi-layer. HIV and Influenza (Fig. 2) are examples of this. The existence of such a lipid bilayer in a virion means that the way it interacts with host cells is somewhat different to virions without a lipid membrane. Importantly, when a virion ‘finds’ a cell to infect, its lipid membrane merges with that of the host cell, inserting the DNA or RNA into the cytosol, the liquid medium inside a cell. Once the viral DNA or RNA has been replicated by the host cell, the virion leaves the cell taking with it a portion of the cell’s membrane. This is explained more fully here.
Figure. 2. Left: coloured image of an Influenza virion; Right: coloured image of an HIV virion. The orange/ochre area of both the Influenza and HIV virions represents that of the lipid surface. Images courtesy of Wikimedia commons and www.topnews.in, respectively.
Such viral ‘budding’ has several interesting features. For example, if the virion needs proteins on its surface (the protruding blobs on the HIV virion, Fig. 2, are an example), those proteins need to be placed there somehow. This requires protein synthesis of the viral proteins by the cell’s machinery. More generally, the lipid profile of the viral membrane is not necessarily that of the cell. So, in order to bud, the virus is reliant on variation in local areas of the cell membrane that have the optimum lipid profile the viral membrane to be constructed.
The idea of a local collection of lipids for virus budding is not inconsistent with that of lipid rafts. There is some debate about how they arise in the context of viral budding, and it is not clear how much direction the virus has in the formation of such areas of local concentration. However, the evidence put forward by Brügger et al. [3] seems clear with respect to which lipids are present in HIV virions. Their work suggests that sphingomyelin lipids are particularly important. This identification of an important lipid for viral production is a valuable molecular target for understanding the behaviour of HIV and also has potential for an approach to HIV therapy. This helps us to understand the manner in which viral replication works.
References
[1] R. Franklin, Nature, 1956, 177, 928-930.
[2] D. L. D. Caspar, Nature, 1956, 177, 928.
[3] B. Brügger, B. Glass, P. Haberkant, I. Leibrecht, F. T. Wieland, H. G. Kräusslich, Proceedings of the National Academy of Sciences, 2005, 103, 2641-2646. www.pnas.org_cgi_doi_10.1073_pnas.0511136103
]]>
Membranes, like everything in cells, need to be controlled and maintained in order to behave in a way that means the individual can remain alive. There is nothing new in this observation, it is a widely-understood and observed process called homeostasis. However, understanding the controls that govern the homeostasis of physical barriers like the membranes of cells is at a much earlier stage.
Once recent development has come from a lab in Southampton, from a group of scientists who have used both lipidomic and physical analyses of the lipid fractions from eukaryotic cells to determine how the membranes of such cells may be managed [1]. One important consideration, and the focus of the study by Dymond et al. is the control of membrane stress.
This may sound trivial or at least a parameter one would assume was kept to a minimum, but rather like the power from a car engine, a certain amount of stress in the membrane is a useful thing. However, this stress is not sufficient to make the membrane bend out of shape spontaneously, and so is known as ‘stored curvature elastic stress’. Instead the stored stress means that when a membrane needs to bend, it can do so in an energetically favoured manner. Such bending processes are vital in the killing off of invading microbes by white blood cells, during the formation of vesicles for transporting neurotransmitters and for cell division.
In order for the curvature elastic stress and the processes themselves to be controlled, the correct lipid building blocks are required. There is good evidence that the lipids present have a dominant influence on the curvature of the assemblies they form, and can even be divided up into ‘types’ on this basis. However, types 0, I and II (Figure 1) seem rather indistinct when we consider how slight curvature might be controlled. A hypothesis based on there being more than just these three types seems more plausible when we consider that lipidomics studies suggest that there are typically several hundred lipid species in even relatively small biological systems [2-5].
Figure 1. Left to right, a type II lipid, inverse cone,e.g.PE; a type 0 lipid, cylinder, e.g.PC; a type I lipid, a normal cone, e.g. lyso-PC.
This has led the Southampton group to research the possibility that there was a universal ‘pivot’ species, i.e., one in between those we currently term type 0 and type II, that appears consistently in all cell lines and growth conditions. The number of lipid species present meant that there were a number of possible candidates, although certain ones, like lyso-PC could be ruled out immediately.
The possible candidates for this pivot species were phosphatidic acid (PA) and phosphatidylethanolamine (PE), the structures of which are shown in Figure 2. The latter may seem an unlikely candidate for a pivot species as it is typically an example of a type II lipid, however, it requires 80% PE to deform a PC system to a type II surface (inverse hexagonal phase). As 80% makes PE the dominant species in that system, it seems reasonable that its power as a type II lipid is not particularly dominating, unlike that of inositide lipids [6,7]. A lipid that is type II in isolation might therefore produce stored curvature elastic stress when mixed with other lipids, especially as a component that is not a majority one.
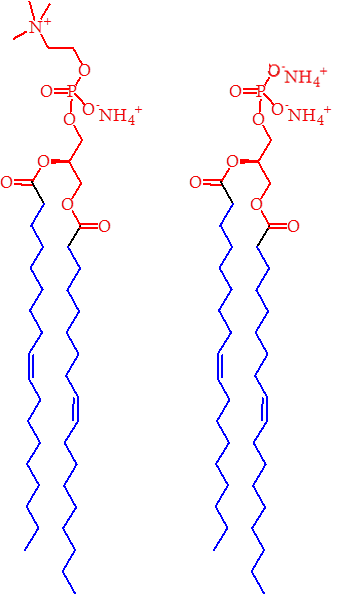
Figure 2. The structures of PE (left) and PA (Right). The blue regions represent the lipophilic part of the lipids, with the red portions representing the hydrophilic parts. On this basis, the head group of PA appears to be smaller than that of PE, suggesting it too might be a type II lipid.
The possibility of a pivot species that is PA is therefore intriguing. It is normally a type 0 lipid, so in order for it to be a pivot species, it must acquire something recognisable as type II character, purely as a way of preventing it remaining simply a type 0 species. The structure of PA (Figure 2) suggests that it has a small and narrow head group, and so it is conceivable that under certain circumstances, that it might exhibit type II morphology (Figure 1), however Dymond et al. report that although this lipid demonstrates some of the character consistent with a pivot species, it does not exhibit the universality required to fit with a general observation. Thus, emphasis fell upon the universal pivot species being a PE lipid.
Some variety of fatty acid residues was also observed to influence the identity of the universal pivot lipid. Although this would be expected to influence only the temperatures at which phase transitions occur, this factor appears important as temperature itself is controlled homeostatically and it seems likely that the two must agree in order to give a stable system.
The natural progression for work such as this is to use it to assess the observations made about lipid compositions in different systems, for example in a comparison of the lipids in cellular membranes of people with obesity-onset diabetes [8].
It therefore seems clear that the presence of a universal pivot species is a useful means for probing both systems under normal, homeostatic conditions, but also for developing an understanding of the physical manifestation of disease, and thus inspire therapies as a result.
References
[1] M. K. Dymond, C. V. Hague, A. D. Postle, G. S. Attard, Journal of the Royal Society – Interface 2013 10, 20120854.
[2] S. Furse, S. Liddell, C. A. Ortori, H. Williams, D. C. Neylon, D. J. Scott, D. A. Barrett, D. A. Gray, Journal of Chemical Biology, 2013, doi:10.1007/s12154-012-0090-1.
[3] E. A. Dennis et al., Journal of Biological Chemistry, 2010, 285, 39976–39985. doi:10.1074/jbc.M110.182915
[4] C.S. Ejsing et al., Proceedings of the National Academy of Science, 2009, 106, 2136–2141. doi:10.1073/pnas.0811700106
[5] O. Quehenberger et al., Journal of Lipid Research, 2010, 51, 3299–3305. doi:10.1194/jlr.M009449
[6] S. Furse, N. J. Brooks, A. M. Seddon, Rudiger Woscholski, R. H. Templer, E. W. Tate, P. R. J. Gaffney and O. Ces, Soft matter, 2012, 8, 3090-3093. doi: 10.1039/c2sm07358g
[7] X. Mulet, R. H. Templer, R. Woscholski and O. Ces, Langmuir, 2008, 24, 8443–8447. doi: 10.1021/la801114n
[8] M. Younsi, D. Quilliot, N. Al-Makdissy, I. Delbachian, P. Drouin, M. Donner, O. Ziegler, Metabolism, 2002, 51, 1261-1268. doi:10.1053/meta.2002.35184