Cholesterol: A Two-Faced Dark Horse?
Cholesterol causes death. At least that is what some people seem to suggest. Or at least if you are a man, apparently it will. There are other examples where it seems to be implicated rather clearly in an alarming context. However, it is equally touted as being instrumental in amazing cures. This rather up-and-down feel to the tabloid portrayal of cholesterol is somewhat at odds with the careful marketing of certain pseudo-dairy products that are “proven to reduce cholesterol”. This latter phrase and related ones imply, of course, that cholesterol is a bad thing.
It is well known that we can have too much of a good thing, especially in our diet. Cholesterol does not quite fall into this category as just about every cell in our body can make all the cholesterol it needs [1]. As it is not a waste product, this suggests to me that it is not overtly harmful: it is obviously important that our bodies have it, otherwise they would not make it. So why is it there? There is a lot of physical evidence that suggests that cholesterol makes up as much as a quarter of the mass of the plasma membranes of several types of human cell [2]. For other cell types, it is less, and for E. Coli bacteria, it is none at all [2]. This presence in cells is partly due to what can be made from cholesterol. It can be made into vitamin D, known as calcipherol (usually written calciferol), a process that famously uses sunlight. It is also the starting material for the body’s production of steroid hormones, the better-known ones being testosterone and the oestrogens. I think these points sound like strong enough reasons for cholesterol to be sloshing around inside us, but there is more.
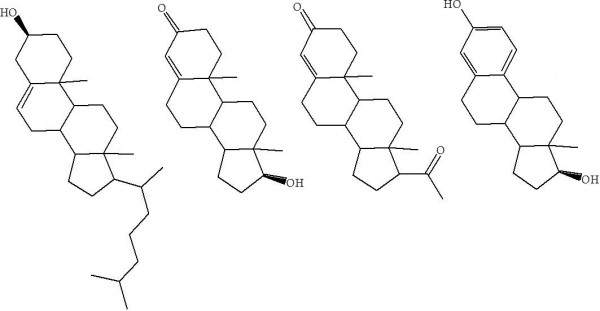
Figure 1. From left, structures of cholesterol, testosterone, progesterone and oestradiol. The latter is one of the three hormones that make up the group of oestrogens.
The utility of cholesterol is not confined to vitamins, sex and puberty. The structure of cholesterol (Figure 1) shows very little functionality that can interact with water, suggesting that it is more lipophilic than hydrophilic. If it cannot dissolve in water, thermodynamic favourability would suggest it is found in fattier parts of the cell. This is indeed the case, with as much as a quarter of the plasma membrane’s mass being cholesterol, it seems reasonable that it must have a physical impact on the properties of the membrane, implying that it must have some purpose. This has indeed been researched, and the results suggest that the presence of cholesterol stiffens the membranes [3]. This is described as a change in the membrane’s fluidity [4], with higher cholesterol giving rise to greater stiffness. Thus cholesterol is part of the system that gives a cell its shape. If you are thinking that most cells are rather shapeless blobs, consider for a moment how different the shape of a sperm cell is to a red blood cell, or an ovum. For some, their shape is very precisely controlled.
The stiffening of the plasma membrane has further ramifications for diffusion across the membrane. Sodium ions, potassium ions and water diffuse more easily across more fluid membranes. Thus the presence of cholesterol influences the speed of diffusion of these particles. These are important things that the cell must keep a check on in order to survive. It is perhaps particularly surprising to note that cholesterol is able to achieve all of these things without being a lipid – it is not amphiphilic. However as it is so closely associated with lipids and lipid systems, it is usually given a place in databases on the subject, as well as in journals on the physical chemistry of lipids.
The fact that cholesterol has its fingers in many bodily pies does rather beg the question of what happens when cholesterol is too high. Physically of course, we can imagine that the membrane will be stiffer than is optimum, reducing the cell’s ability to change shape without being damaged, and with compromised diffusion across the membrane. But what else? The easy answer is what is known as furring of the arteries, so called due to the appearance of the inside wall of the arteries under such circumstances. In reality, no fur has formed. What has happened is in fact physical and is related to the hydrophobic effect. Cholesterol, not being able to dissolve in water, concentrates here along with calcium salts. The latter is a well understood and unrelated phenomenon that is age-related. The suggestion arising from concentration of cholesterol in arteries is, naturally, that too high a level of cholesterol means that there is more to deposit.
This gives a rather nasty sting in the tail for a molecule that is otherwise rather essential for us to be what we are; in fact no vertebrate would be able to reach adulthood without its derivatives. What this story serves to highlight is the double-edged nature of many of the lipid-related biological molecules: not only can we have too much of a good thing, but too much of an essential one as well.
References
[1] F. Xu, S. D. Rychnovsky, J. D. Belani, H. H. Hobbs, J. C. Cohen, and R. B. Rawson, Proc. Natl. Acad. Sci. USA 2005, 102, 14551-14556. http://www.pnas.org/content/102/41/14551.full
[2] P. L. Yeagle, Biochim. et Biophys. Acta, 1985, 822, 267-287. http://www.sciencedirect.com/science/article/pii/0304415785900115
[3] K. E. Bloch CRC Crit. Rev. Biochem., 1983, 14,47-92. http://informahealthcare.com/doi/pdfplus/10.3109/10409238309102790
[4] J. A. Clarke, A. J. Heron, J. M. Seddon, R. V. Law Biophys. J., 2006, 90, 2383-2393. http://www.sciencedirect.com/science/article/pii/S0006349506724213