Fat is a Biological Issue Saturday, Feb 18 2012
One of the first things we may notice when we meet someone new is whether they are fat or thin. Equally, one of the first things we notice about people we know, but may not have seen for some time, is whether they have gained, or lost, weight. The fact that the mass of adipose tissue can differ within an individual, and indeed fluctuate in their lifetime, is perhaps a clue to how our bodies store fats.
It is correctly well known that fats are a biological energy store. In mammals, fats are stored in cells called adipocytes. It is a useful adaptation to be able to store energy for use later, as it means that meals can be further apart and larger, if necessary. Some species are much better at this than us, as demonstrated recently on a BBC documentary by Prof Richard Fortey. Some species of turtle, and crocodiles, can survive for well over six months without a meal. Indeed, this is an important factor in how their species survived a mass extinction event 65 million years ago in which their previous sources of food were compromised. The ability of such cold-blooded creatures to reduce their calorific expenditure, by lowering their body temperature, is also a factor. Although warm-blooded mammals such as humans would be hard-pushed to survive such an event if it happened today, we have at least adapted such that typically, missing a meal or even two, does not result in death due to starvation.
These observations beg questions about how fat is stored in our bodies, how it is stored in the bodies of metabolically efficient reptiles like crocodiles, and how our bodies release these stores of energy. It would be no good if the stored energy were unavailable at the crucial moment. Equally when the energy is released, it needs to be the right amount such that the individual can do what it needs to, without wasting those stores.
Mammals differ from plants in how fat is stored: in mammals there is one large fat droplet per adipocyte, whereas plants tend to use several, smaller stores. Adipocytes can be up to 120 µm wide, roughly 15 times wider than a sperm cell (8 µm) [1]. This enables them to store large amounts of fat in each cell. This is part of the reason why, under normal circumstances, the number of adipocytes in a person does not change. What changes if they gain or lose weight is the size of the fat store in each cell. The operation that removes some of the fat cells is called liposuction.
The vehicle for this storage of energy is the triglyceride (Figure 1). Interestingly, this is exactly the same molecule used by plants for the storage of fat, and is the principal constituent of olive oil. Triglycerides are moved around our bodies by assemblies called lipoproteins. This means that when muscular tissue requires energy from fat stores, it can call upon that stored in adipose tissue, similar to the way that you can order your food shopping on-line and have it delivered to your house.
Once the fat arrives at the cells that will metabolise it, such as muscle or liver cells, the lipoproteins are taken up by the cells and place the fat in small reserves in the cell [2]. These are not convenient for long-term storage in muscle cells in particular, and so the vesicle that contains the lipoprotein is soon joined by another organelle called a lysosome. This organelle provides the enzymes required for metabolising the proteins delivered, while the triglycerides are given over to mitochondria. Mitochondria are responsible for turning the fat into energy and it is thus not for nothing that they are known as the power-houses of the cell.
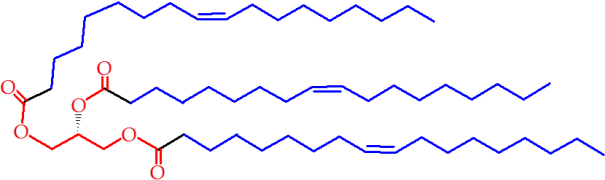
Figure 1. Triglyceride, a molecule composed of three fatty acid molecules and one glycerol moiety. The blue sections represent the lipophilic part of this pseudo-lipid, where the red section is the most polar part of it. However, this polarity is insufficient to confer self-assembly properties on the molecule and thus it is a fat, rather than a lipid.
The chemical process goes by the unlikely-sounding Citric Acid Cycle (CAC), the discovery of which won Sir Hans Krebs a Nobel Prize in 1953.
You may be wondering where the atoms that made up the fats go. Needless to say they are not destroyed by our bodies, so they must be dealt with somehow. Figure 1 shows that fats are made up of carbon, hydrogen and a little bit of oxygen. The hydrogen binds to oxygen, forming water that can either be lost in urine, or more usually, as water vapour in our breath*. This is only part of the reason that heavier breathing is typical during physical exertion; the carbon from fats is turned into carbon dioxide that is also lost when we breathe out.
One last point about fats is that, like cholesterol, we can make as much of it as we need to. Thus the nutritional requirement for fat is virtually nought, though they do have an impact on our enjoyment of a meal. This adaptation indicates to biologists that a varied and not necessarily regular dietary intake was a significant factor at some point in human evolution: we have adapted to enjoy eating fats but can make all we need.
References and Further Reading
[1] B. Alberts, A. Johnson, J. Lewis, M. Raff, K. Roberts, P. Walter, Molecular Biology of the Cell, Garland publishing, 3rd edition, 0815316208.
[2] D. Voet, J. G. Voet, C. W. Pratt, Fundamentals of Biochemistry, John Wiley, 0471586201.
*We lose around 0•1 mL of water each time we breathe out. Based on breathing out once every 3 seconds, this means we breathe out just under 3 L of water each day. Three or four urinations per day, of around 200 mL each, give rise to the loss of less than 1 L of water/day.